As the number two leading cause of death in the US, cancer touches just about everyone in some way. There are many factors involved in the formation of cancer, and genetic changes are a key culprit. Now, a new study sheds light on how the fusion of one normal cell with another can trigger genomic events that turn normal cells cancerous, allowing tumors to form.

According to the American Cancer Society, in the US in 2015, around 1.6 million new cancer cases will be diagnosed, and over 589,000 individuals will die from the disease.
To further investigate causes for this ubiquitous condition, researchers from the University of Michigan and the Mayo Clinic undertook a study that is published in The American Journal of Pathology.
They note that exposure to carcinogens and infectious agents can trigger spontaneous genetic mutations that arise when cells divide. Some researchers, however, believe that the link between carcinogens and genetic changes are too uncertain to account fully for the development of common cancers.
However, researchers have suspected that cell fusion – the process whereby one or more cells combine to form a new cell with multiple nuclei – is a potential cause for some cancers, providing a reason why multiple genetic changes appear to underpin cancer development.
But there has been a lack of clear evidence that normal cell fusion alone could trigger cancer – until now, that is. In their latest study, the researchers show the missing link between cell fusion and the multiple genetic changes that transform normal cells into cancerous cells.
Additionally, the team has been able to show how, when injected into live animals, these fused cells form tumors.
To conduct their study, the researchers used rat IEC-6 intestinal epithelial cells. They explain that these cells maintain a “stable diploid genomic structure” (one with two sets of chromosomes), replicate normally and lack the cellular features of cancer cells. These cells also do not form tumors when observed over several generations.
The team labeled the IEC-6 cells with either red or green dyes and then exposed them to 50% polyethylene glycol to facilitate cell fusion. The researchers could then determine if the cells had fused by noting the presence of both the red and green dyes within one cell.
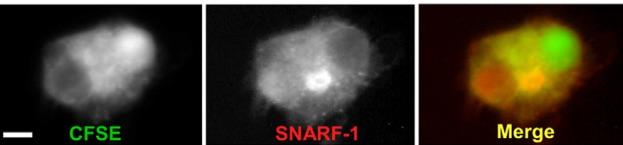
Fused cell image sorted by FACS and visualized by fluorescence microscopy. The fused cell emits both CFSE (green) and SNARF-1 (red) fluorescence.
Image credit: The American Journal of Pathology
While non-fused cells only contained one color, the researchers also noted that fused cells were larger than their non-fused counterparts.
Results reveal that fused cells can replicate – 19% of fused IEC-6 cells generated clones – and with replication, the chromosomes from the two separate cells fused together.
Additionally, the researchers found that 41% of the clones had abnormal numbers of chromosomes, 56% were near-diploid and 4% were tetraploid. In contrast, 86% of the non-fused cells were diploid.
Dr. Jeffrey L. Platt, from the University of Michigan-Ann Arbor, says their results “indicate that cell fusion generates chromosomal instability,” which refers to changes in the appearance and number of chromosomes. Because such abnormalities are typically observed in cancer, the team looked for DNA damage in the fused clones and found such damage in significantly more fused clones than non-fused clones.
They say their findings suggest that after cells fuse, a chromosomal instability might result in DNA damage and, therefore, genetic changes that underpin cancer.
Commenting on their findings, cancer specialist William B. Coleman, PhD, from the University of North Carolina Comprehensive Cancer Center in Chapel Hill, says:
“The frequency of cell fusion events in vivo is not known, although cell fusion is thought to occur under some circumstances such as cell injury, inflammation and viral infection. Although fusion of normal cells in vitro and in vivo may be a rare event, this study shows that cell fusion between normal cells can have pathological consequences.”
He adds that their results “provide evidence for another molecular mechanism driving neoplastic transformation – genomic catastrophe.”
Interestingly, the team also found that when they transplanted IEC-6 cells into immunodeficient mice, during 12 weeks, they generated tumors in 61% of the hosts. In contrast, no tumors formed from the cells that did not fuse.
“We believe one cell fusion event can both initiate malignancy and fuel evolution of the tumor that ensues,” says lead author Xiaofeng Zhou, from the University of Michigan-Ann Arbor.
Dr. Coleman also adds that most cases of human spontaneous cancer are believed to come from cells that underwent random DNA damage or random errors during replication.
He calls for further research to determine whether cell fusion events between normal human cell types can bring about genomic catastrophe and neoplastic transformation.
Last week, Medical News Today reported on two studies that suggested immunotherapy is highly effective against cancer.