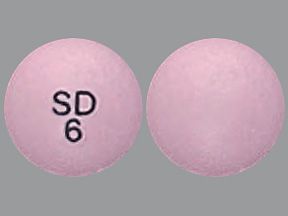
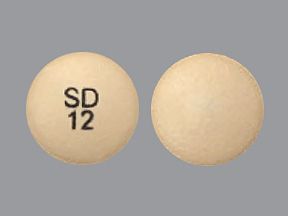
Austedo is a brand-name drug that’s prescribed for certain movement disorders. The medication is available as an oral tablet called Austedo and an extended-release oral tablet called Austedo XR.
Austedo and Austedo XR are approved by the Food and Drug Administration (FDA) to treat the following movement disorders in adults:
- tardive dyskinesia
- chorea related to Huntington’s disease
Note: This article mainly focuses on Austedo. However, the information presented here also applies to Austedo XR unless otherwise noted.
Drug details
You’ll find key information about Austedo below.
- Drug class: vesicular monoamine transporter 2 (VMAT2) inhibitor
- Drug forms: oral tablet, extended-release* oral tablet (Austedo XR)
- Generic available? no
- Prescription required? yes
- Controlled substance? no
- Year of FDA approval: 2017
* Extended release means the drug is slowly released into your body over a long period of time.
Austedo is available only as a brand-name medication. It’s not currently available in generic form. (A generic drug is an exact copy of the active drug in a brand-name medication.)
Austedo contains the active drug deutetrabenazine.
The Austedo or Austedo XR dosage your doctor prescribes will depend on several factors. These include:
- the type and severity of the condition you’re using the drug to treat
- how well your body responds to the drug
- if you have any side effects from this medication
- other medications you may be taking
- if you’re switching to this drug from a different medication
- if you’re switching from Austedo to Austedo XR or vice versa
Typically, your doctor will start you on a low dosage. Then they’ll adjust it over time to reach the amount that’s right for you. Your doctor will ultimately prescribe the smallest dosage that provides the desired effect.
The following information describes dosages that are commonly used or recommended. However, be sure to take the dosage your doctor prescribes for you. Your doctor will determine the best dosage to fit your needs.
For more information on Austedo’s dosages, refer to this article.
Drug forms and strengths
Austedo comes as an oral tablet in three strengths: 6 milligrams (mg), 9 mg, and 12 mg.
Austedo XR comes as an extended-release oral tablet in three strengths: 6 mg, 12 mg, and 24 mg. (Extended release means the drug is slowly released into your body over a long period of time.)
Dosage for tardive dyskinesia
When treating tardive dyskinesia, your starting dosage of Austedo will likely be 6 mg twice daily. (This gives a total daily dose of 12 mg of Austedo.)
If you’re prescribed Austedo XR for this condition, your starting dosage will likely be 12 mg once daily.
The maximum recommended daily dose of Austedo or Austedo XR is 48 mg.
Then your doctor will increase your dosage depending on how well your condition is improving with treatment. Your dosage will also depend on whether you’re having side effects from the drug.
Dosage for chorea related to Huntington’s disease
When treating chorea related to Huntington’s disease, your starting dosage of Austedo will likely be 6 mg twice daily.
If you’re prescribed Austedo XR for this condition, your starting dosage will likely be 12 mg once daily.
Then your doctor will increase your dosage depending on how well your condition is improving with treatment. Your dosage will also depend on whether you’re having side effects from the drug.
The maximum recommended daily dose of Austedo or Austedo XR is 48 mg.
Switching between Austedo and Austedo XR
If your doctor recommends switching from Austedo to Austedo XR or vice versa for your condition, you’ll take the same total daily dose. But whether you take the dose once daily or divide the dose and take it twice daily depends on the form of Austedo you’re prescribed. For example, if you were taking 24 mg of Austedo twice daily, you would take 48 mg of Austedo XR once daily.
What if I miss a dose?
If you miss a dose of Austedo or Austedo XR, take it as soon as you remember, unless it’s almost time for your next dose. If that’s the case, just skip the missed dose and take your next regularly scheduled dose. Don’t take more than one dose of Austedo or Austedo XR at a time.
To help make sure that you don’t miss a dose, try setting a reminder on your phone. A medication timer may be useful, too.
Will I need to use this drug long term?
Austedo and Austedo XR are meant to be used as long-term treatments. If you and your doctor determine that Austedo or Austedo XR is safe and effective for you, you’ll likely take it long term.
Austedo can cause mild or serious side effects. The following lists contain some of the key side effects that may occur while taking Austedo. These lists don’t include all possible side effects.
For more information on the possible side effects of Austedo, refer to this article. You can also talk with your doctor or pharmacist. They can give you tips on how to deal with any side effects that may be bothersome.
The side effects discussed in this section were reported in clinical trials of Austedo. However, the side effects of Austedo XR are expected to be similar to those of Austedo.
Note: The Food and Drug Administration (FDA) tracks side effects of drugs they have approved. If you would like to report to the FDA a side effect you’ve had with Austedo, you can do so through MedWatch.
Mild side effects
Mild side effects of Austedo can include:*
- feeling sleepy or drowsy
- diarrhea
- dry mouth
- fatigue†
- the common cold
- trouble sleeping
- constipation
- anxiety
Most of these side effects may go away within a few days or a couple of weeks. But if they become more severe or don’t go away, talk with your doctor or pharmacist.
* This is a partial list of mild side effects from Austedo. To learn about less common mild side effects, visit Austedo’s prescribing information or talk with your doctor or pharmacist.
† For more information about this side effect, see “Side effect details” below.
Serious side effects
Serious side effects from Austedo aren’t common, but they can occur. Call your doctor right away if you have serious side effects. Call 911 if your symptoms feel life threatening or if you think you’re having a medical emergency.
Serious side effects and their symptoms can include:
- QT prolongation (long QT syndrome), which is a type of atypical heart rhythm that can be very dangerous. Symptoms may include:
- fainting
- seizures
- feeling fluttering in your chest
- Increased level of prolactin, which is a hormone that stimulates your body to make breast milk.* Symptoms may include:
- irregular periods
- loss of libido
- breast pain
- infertility
- Worsening symptoms of Huntington’s disease, when used to treat chorea associated with this condition.
- Feeling restless or being unable to stay still.
* This side effect wasn’t reported in clinical trials of Austedo, but it has occurred with drugs that are very similar to Austedo. Because of this, the manufacturer of Austedo lists the side effect as a precaution for this drug.
Other serious side effects, which are explained in more detail below in “Side effect details,” include:
- allergic reaction
- neuroleptic malignant syndrome (NMS)
- parkinsonism
- severe sedation and fatigue
- depression or thoughts of suicide in people with Huntington’s disease*
* Austedo has a boxed warning from the FDA regarding the risk of depression and thoughts of suicide in people with Huntington’s disease. A boxed warning is the strongest warning the FDA requires. It alerts doctors and patients about drug effects that may be dangerous.
Side effect details
You may wonder how often certain side effects occur with this drug. Here’s some detail on several of the side effects this drug may cause.
Allergic reaction
As with most drugs, some people can have an allergic reaction after taking Austedo. But it’s not known for sure how many people using Austedo have had an allergic reaction to the drug.
Symptoms of a mild allergic reaction can include:
- skin rash
- itchiness
- flushing
A more severe allergic reaction is rare but possible. Symptoms of a severe allergic reaction can include:
- swelling under your skin, typically in your eyelids, lips, hands, or feet
- swelling of your tongue, mouth, or throat
- trouble breathing
Call your doctor right away if you have a severe allergic reaction to Austedo. Call 911 if your symptoms feel life threatening or if you think you’re having a medical emergency.
Neuroleptic malignant syndrome
Neuroleptic malignant syndrome (NMS) is a condition that can occur when you’re using drugs that reduce dopamine levels in your body. (Dopamine is a brain chemical.) Symptoms of NMS can include:
- high fever
- trouble thinking clearly
- increased sweating
- stiff muscles
- very fast or irregular heart rate
In clinical trials of Austedo, there were no reports of NMS. However, this condition has been reported in people taking medications that are similar to Austedo.
If you have any symptoms of NMS while you’re taking Austedo, call your doctor right away. But if your symptoms feel life threatening, call 911. This condition is a medical emergency, and requires treatment as soon as possible.
Parkinsonism
Austedo may cause parkinsonism* in some people. With parkinsonism, you have symptoms that are similar to those caused by Parkinson’s disease. These symptoms include:
- tremor (mild shakiness)
- rigidity (body stiffness)
- trouble moving or keeping your balance
- falling
It’s not known for sure how many people taking Austedo have had parkinsonism. But this side effect has occurred in some people using the drug. In most cases, this happens within the first 2 weeks of starting Austedo or after increasing the dose.
If you have symptoms of parkinsonism while you’re taking Austedo, talk with your doctor. They can recommend whether you need medical treatment. Your doctor may also lower your Austedo dosage or have you stop taking the drug if you develop parkinsonism.
* It’s important to note that parkinsonism is also a possible symptom of Huntington’s disease, which is a condition that Austedo is used to help manage. (Huntington’s disease is a genetic disorder that affects nerves in your brain.)
Severe sedation and fatigue
It’s possible to feel sedated (very sleepy) or fatigued when you’re taking Austedo.
Severe sedation and fatigue are often the side effects of Austedo that prevent your doctor from increasing your dosage of the drug.
While you’re taking Austedo, make sure you don’t drive or use machinery until you know how the drug affects you. And if you feel sedated or very tired during Austedo treatment, talk with your doctor about ways to improve your energy levels.
Depression and thoughts of suicide
Austedo may increase the risk of depression and suicidal thoughts and actions in people with Huntington’s disease. (Huntington’s disease is a genetic condition that affects nerves in your brain.) It’s important to note that people with Huntington’s disease already have an increased risk of depression.
In fact, the Food and Drug Administration (FDA) requires Austedo to have a boxed warning for increased risk of depression and suicidal thoughts or actions. A boxed warning is the strongest warning the FDA can require. It alerts patients and doctors about drug effects that may be dangerous.
Because of these risks, your doctor may not prescribe Austedo if you have depression that’s not fully treated or if you have had or are experiencing suicidal thoughts.
If you have any changes in your mood, behavior, or thoughts while you’re taking Austedo, tell your doctor right away. These symptoms could mean that you have depression. If you develop depression or you have thoughts of suicide while using Austedo, your doctor will recommend a treatment plan that’s right for you.
Suicide prevention
If you know someone at immediate risk of self-harm, suicide, or hurting another person:
- Ask the tough question: “Are you considering suicide?”
- Listen to the person without judgment.
- Call 911 or the local emergency number, or text TALK to 741741 to communicate with a trained crisis counselor.
- Stay with the person until professional help arrives.
- Try to remove any weapons, medications, or other potentially harmful objects.
If you or someone you know is having thoughts of suicide, a prevention hotline can help. The 988 Suicide and Crisis Lifeline is available 24 hours a day at 988. During a crisis, people who are hard of hearing can use their preferred relay service or dial 711 then 988.
As with all medications, the cost of Austedo or Austedo XR can vary.
The actual price you’ll pay depends on your insurance plan, your location, and the pharmacy you use.
Your insurance plan may require you to get prior authorization before approving coverage for Austedo or Austedo XR. This means that your doctor and insurance company will need to communicate about your prescription before the insurance company will cover the drug. The insurance company will review the request and let you and your doctor know whether your plan will cover Austedo.
If you’re not sure whether you’ll need to get prior authorization for Austedo, contact your insurance company.
For more details on Austedo and cost, see this article.
Drug savings
You can explore saving options from the drug manufacturer. (For more options, see the “Financial and insurance assistance” section below.)
Save on your Austedo prescription
Use your insurance to pay as little as $- through Austedo:’s manufacturer savings card.
Financial and insurance assistance
If you need financial support to pay for Austedo or Austedo XR, or if you need help understanding your insurance coverage, help is available.
Teva, the manufacturer of Austedo, offers a program called Shared Solutions, which provides information on cost assistance for these drugs.
For more information and to find out if you’re eligible for support, call 800-887-8100. You can also visit the program website for people with tardive dyskinesia or the program website for people with chorea related to Huntington’s disease.
Generic version
Austedo and Austedo XR aren’t available in generic forms. A generic drug is an exact copy of the active drug in a brand-name medication. Generics tend to cost less than brand-name drugs.
Other drugs are available that can treat your condition. Some may be a better fit for you than others. If you’re interested in finding an alternative to Austedo, talk with your doctor. They can tell you about other medications that may work well for you.
Note: Some of the drugs listed below are used off-label to treat these specific conditions. Off-label use is when a drug that’s approved to treat one condition is used to treat a different condition.
Alternatives for tardive dyskinesia
Examples of other drugs that may be used to treat tardive dyskinesia include:
- valbenazine (Ingrezza)
- clonazepam (Klonopin)
- pramipexole (Mirapex XR)
- ropinirole
- tetrabenazine (Xenazine)
Alternatives for chorea related to Huntington’s disease
Examples of other drugs that may be used to treat chorea related to Huntington’s disease include:
- tetrabenazine (Xenazine)
- haloperidol (Haldol)
- risperidone (Risperdal)
- chlorpromazine
You may wonder how Austedo compares with other medications that are prescribed for similar uses. Here we look at how Austedo and Ingrezza are alike and different.
Ingredients
Austedo contains the active drug deutetrabenazine, while Ingrezza contains the active drug valbenazine. Both medications belong to a class of drugs called vesicular monoamine transporter 2 (VMAT2) inhibitors.
Uses
Both Austedo and Ingrezza are approved to treat the following conditions in adults:
- Tardive dyskinesia: With tardive dyskinesia, you have involuntary, repetitive movements, especially in your face. This condition is related to using certain medications that affect chemicals in your brain.
- Chorea related to Huntington’s disease: With chorea, you have jerking or twitching movements that are involuntary. This disorder has various causes. But these drugs are approved to treat chorea resulting from Huntington’s disease, a genetic condition that affects nerves in your brain.
Drug forms and administration
Austedo comes as an oral tablet that is taken twice daily. It also comes as an extended-release tablet called Austedo XR that is taken once daily. (Extended release means the drug is slowly released into your body over a long period of time.)
Ingrezza comes as an oral capsule that is taken once daily.
Side effects and risks
Austedo and Ingrezza both contain active drugs that belong to the same class of drugs. Therefore, these medications can cause very similar side effects. Below are examples of these side effects.
Mild side effects
These lists contain up to 10 of the most common mild side effects that can occur with each drug, or with both Austedo and Ingrezza (when taken individually).
- Can occur with Austedo:
- diarrhea
- the common cold
- trouble sleeping
- Can occur with Ingrezza:
- changes in balance or problems keeping your balance
- headache
- blurry vision
- Can occur with both Austedo and Ingrezza:
- fatigue
- feeling sleepy or drowsy
- dry mouth
- constipation
Serious side effects
These lists contain examples of serious side effects that can occur with Austedo, with Ingrezza, or with both drugs (when taken individually).
- Can occur with Austedo:
- no unique serious side effects
- Can occur with Ingrezza:
- no unique serious side effects
- Can occur with both Austedo and Ingrezza:
- QT prolongation (long QT syndrome), which is a type of atypical heart rhythm that can be very dangerous
- severe sedation or fatigue
- parkinsonism (movement-related symptoms that are similar to those caused by Parkinson’s disease)
- depression and suicidal thoughts or actions in people with Huntington’s disease*
- neuroleptic malignant syndrome (NMS), which is a condition that’s caused by a chemical imbalance in your brain
- feeling restless or being unable to stay still
* Austedo and Ingrezza have a boxed warning from the FDA regarding the risk of depression and thoughts of suicide in people with Huntington’s disease. A boxed warning is the strongest warning the FDA requires. It alerts doctors and patients about drug effects that may be dangerous.
Effectiveness
Austedo and Ingrezza are both used to treat tardive dyskinesia and chorea related to Huntington’s disease in adults.
These drugs haven’t been directly compared in clinical trials. But separate clinical trials have found both Austedo and Ingrezza to be effective for treating the conditions they are approved for.
Costs
Austedo and Ingrezza are both brand-name drugs. There are currently no generic forms of either drug. Brand-name medications usually cost more than generics.
The actual price you’ll pay for either drug depends on your prescribed dosage, your insurance plan, your location, and the pharmacy you use.
Like Ingrezza (discussed above), other medications are also prescribed for uses similar to those of Austedo. Here we look at how Austedo and Xenazine are alike and different.
Ingredients
Austedo contains the active drug deutetrabenazine, while Xenazine contains the active drug tetrabenazine. Both medications belong to a class of drugs called vesicular monoamine transporter 2 (VMAT2) inhibitors.
Uses
Both Austedo and Xenazine are approved for use in adults to treat chorea related to Huntington’s disease. With chorea, you have jerking or twitching movements that are involuntary. These movements can be caused by Huntington’s disease, which is a condition that affects nerves in your brain.
In addition, Austedo is also approved to treat tardive dyskinesia in adults. With tardive dyskinesia, you have involuntary, repetitive movements, especially in your face.
Drug forms and administration
Austedo comes as an oral tablet that is taken twice daily. It also comes as an extended-release tablet called Austedo XR that is taken once daily. (Extended release means the drug is slowly released into your body over a long period of time.)
Xenazine comes as an oral tablet that is taken up to three times daily.
Side effects and risks
Austedo and Xenazine both belong to the same class of drugs. Therefore, these medications can cause very similar side effects. Below are examples of these side effects.
Mild side effects
These lists contain up to 10 of the most common mild side effects that can occur with each drug, or with both Austedo and Xenazine (when taken individually).
- Can occur with Austedo:
- diarrhea
- dry mouth
- Can occur with Xenazine:
- nausea
- vomiting
- difficulty keeping your balance
- Can occur with both Austedo and Xenazine:
- feeling sleepy or drowsy
- fatigue
- trouble sleeping
- anxiety
- the common cold
Serious side effects
These lists contain examples of serious side effects that can occur with Austedo, with Xenazine, or with both drugs (when taken individually).
- Can occur with Austedo:
- no unique serious side effects
- Can occur with Xenazine:
- reduced blood pressure, which can lead to dizziness or fainting
- Can occur with both Austedo and Xenazine:
- depression and suicidal thoughts or actions in people with Huntington’s disease*
- QT prolongation (long QT syndrome), which is a type of atypical heart rhythm that can be very dangerous
- severe sedation or fatigue
- parkinsonism (movement-related symptoms that are similar to those caused by Parkinson’s disease )
- neuroleptic malignant syndrome (NMS), which is a condition that’s caused by a chemical imbalance in your brain
- feeling restless or being unable to stay still
- increased level of prolactin, which is a hormone that stimulates your body to make breast milk
- worsening symptoms of Huntington’s disease, when used to treat chorea associated with this condition
* Austedo and Xenazine each have a boxed warning from the Food and Drug Administration (FDA) regarding the risk of depression and suicidal thoughts or actions in people with Huntington’s disease. A boxed warning is the strongest warning the FDA requires. It alerts doctors and patients about drug effects that may be dangerous.
Effectiveness
Austedo and Xenazine have different approved uses, but they’re both used in adults to treat chorea related to Huntington’s disease.
These drugs haven’t been directly compared in clinical trials. But separate clinical trials have found both Austedo and Xenazine to be effective for treating chorea related to Huntington’s disease.
Costs
Austedo and Xenazine are both brand-name drugs. There is currently no generic form of Austedo. A generic form of Xenazine, called tetrabenazine, is available. Brand-name medications usually cost more than generics.
The actual price you’ll pay for either Austedo or Xenazine depends on your insurance plan, your location, and the pharmacy you use.
The Food and Drug Administration (FDA) approves prescription drugs such as Austedo and Austedo XR to treat certain conditions. Austedo and Austedo XR may also be used off-label for other conditions. Off-label use is when a drug that’s approved to treat one condition is used to treat a different condition.
Austedo for tardive dyskinesia
Austedo and Austedo XR are FDA-approved to treat tardive dyskinesia in adults. With this condition, you have involuntary, repetitive movements. These movements usually occur in your face, neck, and tongue. But the movements can also happen in your arms, legs, or other parts of your body.
Tardive dyskinesia is caused by using certain medications that block receptors (attachment sites) for the brain chemical dopamine. This causes dopamine to build up to atypically high levels around certain nerves in your body.
Examples of medications that may lead to tardive dyskinesia include:
- drugs used to treat mood disorders, including schizophrenia or depression, such as:
- fluoxetine (Prozac)
- trazodone
- haloperidol (Haldol)
- chlorpromazine
- drugs used to treat Parkinson’s disease, such as levodopa/carbidopa (Sinemet)
- drugs used to treat seizures, such as:
- phenobarbital
- phenytoin (Dilantin)
- metoclopramide (Reglan), which is a drug that’s commonly used to treat nausea and vomiting, and certain other stomach problems
Austedo and Austedo XR work to treat tardive dyskinesia by reducing dopamine levels and stopping dopamine from stimulating certain nerves in your brain.
Effectiveness for tardive dyskinesia
In clinical trials, Austedo was found to be effective in treating tardive dyskinesia. For information about these clinical trials, see Austedo’s prescribing information.
Austedo for chorea related to Huntington’s disease
Austedo and Austedo XR are also FDA-approved for use in adults with chorea related to Huntington’s disease. With chorea, you have involuntary, jerking movements that usually occur in your shoulders, hips, and face.
These movements can be caused by Huntington’s disease, which is a genetic disease that causes nerve cells in your brain to break down. The disease can affect the function of your brain, as well as your ability to control movements.
Chorea may be caused by a buildup of the brain chemical dopamine around certain nerves in your body. Austedo and Austedo XR work to treat chorea by reducing dopamine levels and stopping dopamine from stimulating certain nerves in your brain.
Effectiveness for chorea related to Huntington’s disease
In clinical trials, Austedo was found to be effective in treating chorea related to Huntington’s disease. For information about these clinical trials, see Austedo’s prescribing information.
Austedo for other conditions
In addition to the uses listed above, you may wonder whether Austedo is used for certain other conditions. Below is information on use for Austedo that’s currently being studied.
Austedo for Tourette syndrome (under study)
Austedo and Austedo XR are not FDA-approved to treat Tourette syndrome. Studies evaluating Austedo for this use have not found the drug effective for treating symptoms of Tourette syndrome. If you’d like to know more about treatment options for Tourette syndrome, talk with your doctor.
You should take Austedo according to a doctor or healthcare professional’s instructions.
When to take
Austedo comes as an oral tablet, which you take by mouth twice daily. Austedo XR comes as an extended-release oral tablet, which you take by mouth once daily. (Extended release means the drug is slowly released into your body over a long period of time.)
If you’re taking Austedo, try to take your doses about 12 hours apart. To do this, you could take your first dose in the morning and your second dose in the evening.
To help make sure that you don’t miss a dose, try setting a reminder on your phone. A medication timer may be useful, too.
Taking Austedo with food
You should take Austedo with food.
You can take Austedo XR with or without food.
Can Austedo be crushed, split, or chewed?
No, you shouldn’t crush, chew, or break Austedo or Austedo XR tablets. You should swallow them whole.
Austedo and Austedo XR are approved to treat involuntary movements caused by:
- tardive dyskinesia
- chorea related to Huntington’s disease
The involuntary movements caused by these conditions occur when there’s a buildup of the brain chemical dopamine around certain nerves in the body. For more information on these conditions, see the “Austedo uses” section above.
Austedo and Austedo XR contain the active drug deutetrabenazine. It belongs to a class of drugs called vesicular monoamine transporter 2 (VMAT2) inhibitors. Austedo and Austedo XR work to reduce both chorea and tardive dyskinesia symptoms by decreasing the level of dopamine in your body. This stops dopamine from stimulating certain nerves and causing involuntary movements.
How long does it take to work?
You may notice Austedo starting to work for you after a few weeks of taking the drug. Austedo XR is expected to take a similar amount of time to start working.
In clinical trials, Austedo’s effectiveness in reducing involuntary movements caused by either chorea or tardive dyskinesia was measured using certain scoring systems. The scores took into account involuntary movements that occurred in people’s faces or other areas of their bodies.
People’s movements were measured 2 weeks after their first dose of Austedo. These scores were compared to scores that were recorded before treatment was started. After just 2 weeks of treatment, people’s symptom scores were already lowered.
And by the end of each 12-week trial, people’s symptom scores were lowered even further as their dosage of Austedo was increased.
Drinking alcohol while you’re using Austedo or Austedo XR may increase your risk of feeling sleepy or drowsy. (These are also side effects caused by Austedo and Austedo XR.) Because of this risk, you shouldn’t drink alcohol during treatment with either form of the drug.
If you have questions about the safety of drinking alcohol while using Austedo or Austedo XR, talk with your doctor or pharmacist.
Austedo can interact with several other medications. It can also interact with certain supplements as well as certain foods.
Different interactions can cause different effects. For instance, some interactions can interfere with how well a drug works. Other interactions can increase side effects or make them more severe.
Note: Austedo XR is expected to have the same interactions as the ones described for Austedo in this section.
Austedo and other medications
Below are lists of medications that can interact with Austedo. These lists do not contain all drugs that may interact with Austedo.
Before taking Austedo, talk with your doctor and pharmacist. Tell them about all prescription, over-the-counter, and other drugs you take. Also tell them about any vitamins, herbs, and supplements you use. Sharing this information can help you avoid potential interactions.
If you have questions about drug interactions that may affect you, ask your doctor or pharmacist.
Austedo and certain mood disorder drugs
Some drugs used to treat mood disorders can interact with Austedo. These interactions are described below. If you’re taking any of the drugs listed below, be sure to talk with your doctor before starting Austedo.
Austedo and MAOIs
You should not use Austedo with drugs that belong to a class called monoamine oxidase inhibitors (MAOIs). MAOIs are used to treat certain mood disorders, including depression.
Examples of MAOIs include:
- rasagiline (Azilect)
- selegiline (Zelapar)
- isocarboxazid (Marplan)
- phenelzine (Nardil)
- tranylcypromine (Parnate)
If you take Austedo and an MAOI together, you may have dangerously high blood pressure.
If you have taken or are currently taking an MAOI, make sure you wait at least 14 days after you stop taking the MAOI before you start taking Austedo.
Austedo and mood disorder drugs that increase Austedo levels in your body
Some drugs that are used for mood disorders can increase the level of Austedo in your body. This may increase your risk of side effects from Austedo. Examples of these drugs include:
- paroxetine (Paxil)
- fluoxetine (Prozac)
- bupropion (Wellbutrin SR, Wellbutrin XL)
If you’re taking one of the drugs listed above, your doctor may lower your dosage of Austedo to help you avoid increased side effects from Austedo.
Austedo and mood disorder drugs called dopamine antagonists or antipsychotics
Some drugs used for mood disorders that are called either dopamine antagonists or antipsychotics may interact with Austedo. (Dopamine antagonists block the activity of dopamine, which is a chemical in your brain.)
Examples of these mood disorder drugs include:
- chlorpromazine
- haloperidol (Haldol)
- thioridazine
- ziprasidone (Geodon)
Some of these drugs may interfere with your heart’s electrical activity. Austedo also may affect your heart’s electrical activity. Taking these drugs with Austedo can increase your risk of QT prolongation (long QT syndrome). (QT prolongation is type of atypical heart rhythm that can be dangerous.)
In addition, some of these drugs may increase the risk of feeling restless or being unable to stay still. The drugs may also increase the risk of parkinsonism* and neuroleptic malignant syndrome (NMS).† These risks are also increased by Austedo. Taking these drugs with Austedo may increase these side effects.
If you’re taking one of the drugs listed above, your doctor may lower your dosage of Austedo to help you avoid prolonged QT interval and restlessness.
* With parkinsonism, you have movement-related symptoms that are similar to those caused by Parkinson’s disease.
† NMS is a condition that can occur when you’re using drugs that reduce dopamine levels in your body. (Dopamine is a brain chemical.)
Austedo and certain antibiotics
Some antibiotics may interfere with your heart’s electrical activity. Austedo also may affect your heart’s electrical activity. Taking these drugs together can increase your risk of QT prolongation.
Examples of antibiotics that may affect your heart’s electrical activity include:
- moxifloxacin
- azithromycin (Zithromax)
- clarithromycin
If you’re taking any of the antibiotics listed above, be sure to talk with your doctor before starting Austedo.
Austedo and certain heart medications
Some drugs that are used to treat certain heart conditions may interfere with your heart’s electrical activity. Austedo also may affect your heart’s electrical activity. Taking these drugs together may increase your risk of QT prolongation.
Examples of certain heart medications that may affect your heart’s electrical activity include:
- amiodarone
- sotalol (Betapace)
- quinidine
- procainamide
If you’re taking any of the drugs listed above, be sure to talk with your doctor before starting Austedo.
Austedo and other drugs that are used for chorea or tardive dyskinesia
You shouldn’t take Austedo with other drugs that both:
- belong to the same class of drugs called vesicular monoamine transporter 2 (VMAT2) inhibitors, and
- are used to treat either tardive dyskinesia or chorea related to Huntington’s disease
These drugs include tetrabenazine (Xenazine) and valbenazine (Ingrezza).
If you’re taking either of the drugs mentioned above, be sure to talk with your doctor before starting Austedo.
Austedo and herbs and supplements
There aren’t any herbs or supplements that have been specifically reported to interact with Austedo. However, you should still check with your doctor or pharmacist before using any of these products while taking Austedo.
Austedo and foods
There aren’t any foods that have specifically been reported to interact with Austedo.
There’s not enough information available to know whether it’s safe to use Austedo or Austedo XR during pregnancy. In fact, there haven’t been any studies of this drug during pregnancy.
In animal studies, Austedo increased the risk of stillbirth and newborn deaths due to fetal exposure to the drug. But keep in mind that animal studies don’t always predict what will happen in people.
If you’re pregnant or considering becoming pregnant, talk with your doctor about taking Austedo or Austedo XR. They can discuss with you other treatment options that may be safer when used during pregnancy.
It’s not known whether Austedo or Austedo XR is safe to take during pregnancy. If you’re sexually active and you or your partner can become pregnant, talk with your doctor about your birth control needs while you’re using either of these drugs.
It’s unknown whether it’s safe to take Austedo or Austedo XR while you’re breastfeeding. There haven’t been any studies done of people taking Austedo while breastfeeding. And there also haven’t been any studies done in lactating animals. Because of this, it’s not known whether Austedo or Austedo XR passes into breast milk or what effect these drugs may have on a child who’s breastfed.
If you’re breastfeeding or considering breastfeeding, talk with your doctor before taking Austedo or Austedo XR. They can recommend other treatment options that may be safer to use while you’re breastfeeding. Or they may recommend safe and healthy alternatives for feeding your child.
Here are answers to some frequently asked questions about Austedo.
Note: The information given in this section also applies to Austedo XR.
Will Austedo cure my condition?
No, Austedo is approved to treat two conditions, but it won’t cure them. These conditions are:
- Tardive dyskinesia: With tardive dyskinesia, you have involuntary, repetitive movements, especially in your face. This condition is related to using certain medications that block dopamine receptors (attachment sites). This leads to dopamine building up around certain nerves in your body.
- Chorea related to Huntington’s disease: With chorea, you have jerking or twitching movements that are involuntary. This condition is a symptom of Huntington’s disease, which is a genetic disease that causes the breakdown of nerve cells in your brain. Chorea may be caused by a buildup of the brain chemical dopamine around certain nerves in your body.
Austedo works to treat these conditions by reducing the level of dopamine in your body. This stops dopamine from stimulating certain nerves and causing involuntary movements.
So while Austedo will improve chorea and tardive dyskinesia symptoms, the drug doesn’t cure the root cause of these conditions. To learn more about the cause of tardive dyskinesia and chorea related to Huntington’s disease, see the “Austedo uses” section above.
If you have questions about what you can expect from treatment with Austedo, talk with your doctor.
If I’m taking an antipsychotic, can I keep taking it while I’m using Austedo?
It depends. Certain antipsychotic drugs can’t be taken with Austedo because doing so may cause some dangerous side effects. These side effects include conditions such as QT prolongation (long QT syndrome). QT prolongation is a type of irregular heart rhythm.
Examples of antipsychotics that can increase the risk of QT prolongation if taken with Austedo include:
- chlorpromazine
- haloperidol (Haldol)
Other drugs used to treat mood disorders that may also increase the risk of QT prolongation if taken with Austedo include:
For information about other drugs that may not be safe to use with Austedo, see the “Austedo interactions” section above. And be sure to talk with your doctor about all medications you’re using before you start taking Austedo. They can recommend whether it’s safe to take Austedo with certain other drugs.
Will I have withdrawal symptoms if I stop taking Austedo?
No, you won’t have withdrawal symptoms if you stop taking Austedo. (Withdrawal symptoms can occur when you stop taking a drug that your body has become dependent upon. With drug dependence, your body needs the drug to feel like it usually does.)
However, the condition that you were using Austedo to treat may return. If that happens, you may have increased symptoms of that condition.
Is it safe for older adults to take Austedo?
It’s unknown whether older adults respond differently than younger adults to Austedo. This is because clinical trials that looked at the safety and effectiveness of Austedo didn’t include very many people who were ages 65 years and older.
If you’re age 65 years or older and you’d like to use Austedo, talk with your doctor. If they recommend that you use Austedo, they’ll likely have you start taking a very low dosage. This helps them ensure that you’re doing well on the drug before they increase your dosage.
This drug comes with several precautions. The Austedo precautions described in this section also apply to Austedo XR.
FDA warning: Risk of depression and suicide
This drug has a boxed warning. This is the most serious warning from the Food and Drug Administration (FDA). A boxed warning alerts doctors and patients about drug effects that may be dangerous.
Austedo can increase the risk of depression and suicidal thoughts or actions in people with Huntington’s disease. (Huntington’s disease is a genetic condition that affects nerves in your brain.) This risk is increased in people who have a history of depression and in people who’ve attempted suicide in the past. Because of these risks, your doctor may not prescribe Austedo if you have depression that’s not fully treated or if you have had or are experiencing suicidal thoughts.
Other precautions
Before taking Austedo, talk with your doctor about your health history. Austedo may not be right for you if you have certain medical conditions or other factors affecting your health. These include:
QT prolongation: Austedo may affect your heart’s electrical signaling and cause a condition called QT prolongation (long QT syndrome). This condition can cause an irregular heart rhythm. If you already have certain heart problems, such as an irregular heart rhythm, Austedo may worsen your condition. In addition, taking certain medications with Austedo may also cause new or worsening problems with your heart rhythm.
Long QT syndrome often doesn’t have any symptoms, but sometimes it can be very dangerous. Your doctor will monitor your heart’s electrical activity during Austedo treatment if you have a history of long QT syndrome or if you have symptoms of an irregular heart rhythm.
Liver problems: Your doctor likely won’t prescribe Austedo if you have a liver problem, such as cirrhosis. Your body depends on the liver to break down Austedo after you take a dose. If you have a liver problem, your body may not break down Austedo as well as it should. This can increase your risk of side effects that Austedo may cause, which could be severe.
Low blood potassium or magnesium level: If you have a low blood potassium or low blood magnesium level, you may be at higher risk for long QT syndrome as a side effect of Austedo. Your doctor can discuss whether Austedo is safe for you to take. They may want to treat your low blood potassium or magnesium level first before prescribing Austedo.
Pregnancy: There’s not enough information available to know whether it’s safe to use Austedo during pregnancy. For more information, see the “Austedo and pregnancy” section above.
Breastfeeding: It’s not known whether Austedo is safe to take while breastfeeding. For more information, see the “Austedo and breastfeeding” section above.
Note: For more information about the potential negative effects of Austedo, see the “Austedo side effects” section above.
Using more than the recommended dosage of Austedo or Austedo XR can lead to serious side effects. Don’t use more of either drug than your doctor recommends.
Overdose symptoms
The manufacturer of Austedo and Austedo XR has not reported overdose symptoms for either drug. However, overdose symptoms for these drugs are likely to resemble those of a similar drug, tetrabenazine (Xenazine). These overdose symptoms include:
- nausea or vomiting
- involuntary muscle spasms
- your eyes rolling upward and back
- sweating
- tiredness
- low blood pressure
- confusion
- diarrhea
- hallucinations (seeing or hearing things that aren’t really there)
- tremors
- skin discoloration
What to do in case of overdose
If you think you’ve taken too much of this drug, call your doctor. You can also call America’s Poison Centers at 800-222-1222 or visit its website. But if your symptoms are severe, call 911 or go to the nearest emergency room right away.
When you get Austedo from the pharmacy, the pharmacist will add an expiration date to the label on the bottle. This date is typically 1 year from the date they dispensed the medication.
The expiration date helps guarantee that the medication is effective during this time. The
Storage
How long a medication remains good can depend on many factors, including how and where you store the medication.
Austedo and Austedo XR tablets should be stored at a room temperature of 77°F (25°C) in a tightly sealed container away from light. Avoid storing this medication in areas where it could get damp or wet, such as bathrooms.
Disposal
If you no longer need to take Austedo and have leftover medication, it’s important to dispose of it safely. This helps prevent others, including children and pets, from taking the drug by accident. It also helps keep the drug from harming the environment.
This article provides several useful tips on medication disposal. You can also ask your pharmacist for information on how to dispose of your medication.
Disclaimer: Medical News Today has made every effort to make certain that all information is factually correct, comprehensive, and up to date. However, this article should not be used as a substitute for the knowledge and expertise of a licensed healthcare professional. You should always consult your doctor or another healthcare professional before taking any medication. The drug information contained herein is subject to change and is not intended to cover all possible uses, directions, precautions, warnings, drug interactions, allergic reactions, or adverse effects. The absence of warnings or other information for a given drug does not indicate that the drug or drug combination is safe, effective, or appropriate for all patients or all specific uses.