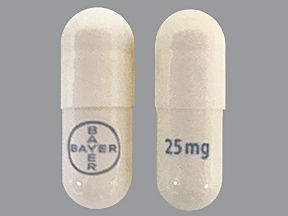
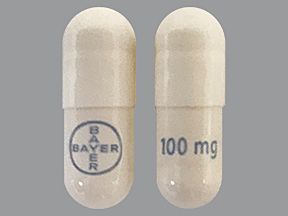
Vitrakvi (larotrectinib) is a brand-name drug that’s prescribed for solid tumors in certain situations. The medication is available in the following forms: oral capsule and solution. Vitrakvi belongs to the kinase inhibitors drug class.
Vitrakvi is FDA-approved to treat solid tumors (cancer) that meet the following conditions:
- have certain genes that have fused together abnormally, and
- are metastatic (have spread) or can’t be removed through surgery because it could cause serious complications, and
- spread after you’ve tried other treatments or other effective treatments aren’t available
Vitrakvi is approved for use in adults and children. (For more information about the uses of Vitrakvi, see the “Vitrakvi uses” section below.)
FDA approval
Here’s some information on the approval of Vitrakvi by the Food and Drug Administration (FDA).
Accelerated approval
In 2018, the FDA granted Vitrakvi
Accelerated approval is reserved for certain drugs that treat conditions without a lot of effective treatment options. For example, if you have the type of solid tumor mentioned above and surgery isn’t safe, there aren’t many effective treatments available.
The FDA won’t decide whether to give Vitrakvi full approval until after more clinical trials have been completed. Trials are still being conducted.
Orphan drug status
Vitrakvi has also received
Orphan drug status means the medication is approved to treat certain rare conditions that don’t have many or any treatment options. (The type of solid tumor Vitrakvi treats is considered rare.) The goal of the status is to encourage more research into the rare conditions.
If a medication has been given orphan drug status, clinical trials have shown that the drug is safe and effective. This is the same clinical trial process for other medications approved by the FDA.
Effectiveness
For information about the effectiveness of Vitrakvi, see the “Vitrakvi uses” section below.
As with all medications, the cost of Vitrakvi can vary.
Visit this page to access Optum Perks coupons and get price estimates for Vitrakvi when you use the coupons. These coupons can provide significant savings on your prescription costs.
Note: Optum Perks coupons cannot be used with any insurance copays or benefits.
The actual price you’ll pay depends on your insurance plan, your location, and the pharmacy you use.
Keep in mind that you may be able to get a 90-day supply of Vitrakvi. If approved by your insurance company, getting a 90-day supply of the drug could reduce your number of trips to the pharmacy and help lower the cost. If you’re interested in this option, check with your doctor or your insurance company.
It’s important to note that you’ll have to get Vitrakvi at a specialty pharmacy. This type of pharmacy is authorized to carry specialty medications. These are drugs that may be expensive or may require help from healthcare professionals to be used safely and effectively.
Before approving coverage for Vitrakvi, your insurance company may require you to get prior authorization. This means that your doctor and insurance company will need to communicate about your prescription before the insurance company will cover the drug. The insurance company will review the prior authorization request and decide if the drug will be covered.
If you’re not sure if you’ll need to get prior authorization for Vitrakvi, contact your insurance company.
Financial and insurance assistance
If you need financial support to pay for Vitrakvi, or if you need help understanding your insurance coverage, help is available.
A patient assistance program is available for Vitrakvi, which can help you with financial assistance for this drug. For more information and to find out if you’re eligible for support, call 800-288-8374 or visit the program website.
Mail-order pharmacies
Vitrakvi may be available through a mail-order specialty pharmacy. Using this service may help lower the drug’s cost and allow you to get your medication without leaving home.
If recommended by your doctor, you may be able to receive a 90-day supply of Vitrakvi, so there’s less concern about running out of the medication. If you’re interested in this option, check with your doctor and your insurance company. Some Medicare plans may help cover the cost of mail-order medications.
If you don’t have insurance, you can ask your doctor or pharmacist about online pharmacy options.
Generic version
Vitrakvi isn’t available in a generic form. A generic drug is an exact copy of the active drug in a brand-name medication. Generics tend to cost less than brand-name drugs.
Vitrakvi is available only as a brand-name medication. It’s not currently available in generic form.
A generic drug is an exact copy of the active drug in a brand-name medication. Generics usually cost less than brand-name drugs.
The active drug in Vitrakvi is larotrectinib.
The Food and Drug Administration (FDA) approves prescription drugs such as Vitrakvi to treat certain conditions.
Vitrakvi for solid tumors
Vitrakvi is FDA-approved to treat solid tumors (cancer) that meet the following conditions:
- The tumors must have genes called neurotrophic receptor tyrosine kinases that have fused together abnormally. This is known as gene fusion. The fused genes must not have certain mutations (changes) that could make the tumor resistant* to treatment with Vitrakvi. Before taking Vitrakvi, your doctor will perform a test to find out whether your tumor carries these genes and does not have those mutations.
- Also, the tumors must be metastatic (have spread) or surgery to remove the tumors could cause serious complications.
- In addition, the tumors must have spreadafter you tried other treatments or other effective treatments aren’t available.
Vitrakvi is approved for use in adults and children.
* A tumor that’s resistant to treatment is one that’s harder to treat than usual or can’t be treated.
Solid tumors explained
Solid tumors are abnormal clumps of cells. Typically, cells in your body grow, divide, and replace each other. Old cells die as new cells are made. Some tumors are benign, meaning they aren’t cancerous, but others are malignant (cancerous). When a person has cancer, new cells divide more than they should or old cells don’t die when they should.
Symptoms of solid tumors can vary based on where the tumor occurs in the body. But there are some general symptoms that are the same with different solid tumors. Solid tumors often cause swelling or a mass that you can feel. Other general symptoms can include weight loss, fever, and a vague feeling of being ill or sick.
Examples of the different types of cancers involving solid tumors include:
Some cancer medications treat tumors depending on where in the body they’re located. This isn’t the case with Vitrakvi. Instead, the tumors must have special genetic markers that your doctor can look for. The tumors also need to be solid. Vitrakvi isn’t approved to treat cancers of the blood, such as leukemia.
For more information about the conditions Vitrakvi is used to treat, see our cancer hub.
Effectiveness for solid tumors
Vitrakvi’s effectiveness in treating solid tumors was shown across three clinical trials. None of these trials compared Vitrakvi with a different cancer drug or a placebo (treatment with no active drug). Adults and children in the trials received Vitrakvi until their tumor progressed (got worse) or they had side effects that were bothersome to them.
Partial and complete response rates
Researchers wanted to see if people’s tumors responded to Vitrakvi, so they measured partial and complete response rates. With a partial response, the treatment lowered the level of cancer cells to some degree. With a complete response, the treatment lowered the level of cancer cells so much that imaging tests couldn’t detect the cells. Examples of imaging tests include X-rays, CT scans, and MRI scans.
The researchers found that:
- 49% of people had a partial response to Vitrakvi
- 25% of people had a complete response to Vitrakvi
Of people who had either a partial or complete response, the response lasted for:
- at least 12 months in 63% of people
- at least 24 months in 49% of people
Overall response rate
When looking at cancer treatments, researchers can also measure the overall response rate. This is how many people had either a partial or complete response to a treatment.
Here are some* of the overall response rates from the trials mentioned above:
| Cancer type | Number of people enrolled | Overall response rate |
| Colon | 4 | 25% |
| Lung | 4 | 75% |
| Melanoma | 4 | 50% |
| Salivary gland | 12 | 83% |
| Soft tissue† | 11 | 91% |
| Thyroid | 5 | 100% |
* For full clinical trial results, including a list of all of the solid tumor types treated by Vitrakvi in these trials, see the drug’s prescribing information. More research is needed to understand how Vitrakvi’s effect on response rate varies between cancer types.
† “Soft tissue” refers to cancers that start in supportive tissue, such as muscle, fat, nerves, or tendons.
Vitrakvi and children
Vitrakvi is FDA-approved to treat children with solid tumors (cancer) that meet certain conditions. For details, see “Vitrakvi for solid tumors” above.
In clinical studies, Vitrakvi was given to children ages 1 month and older.
Researchers in these studies weren’t able to separate data based on age. But they did look at seven children with infantile fibrosarcoma. All of these children had an overall response to Vitrakvi. An overall response is how many people had either a partial or complete response to a treatment.
With a partial response, the treatment lowered the level of cancer cells to some degree. With a complete response, the treatment lowered the level of cancer cells so much that blood tests couldn’t detect the cells.
The Vitrakvi dosage your doctor prescribes will depend on several factors. These include:
- your age, body weight, and body surface area
- side effects you may have while taking the drug
- other medical conditions you may have
- other medications you may be taking
The following information describes dosages that are commonly used or recommended. However, be sure to take the dosage your doctor prescribes for you. Your doctor will determine the best dosage to fit your needs.
Drug forms and strengths
Vitrakvi comes in two forms:
- Capsules. You’ll swallow the capsules. They’re available in two strengths: 25 milligrams (mg) and 100 mg.
- Liquid solution. You’ll use an oral syringe to swallow the solution. It’s available in one strength: 20 mg per milliliter (mL).
You’ll likely be able to take either form of the drug.
Dosage for solid tumors
The recommended Vitrakvi dosage for treating solid tumors (cancer) in adults is 100 mg twice daily.
If your tumor grows or if you have side effects that you can’t tolerate, your doctor may lower your dosage. Or they may have you stop taking the drug.
Children’s dosage
The Vitrakvi dosage for treating solid tumors in children is based on the child’s body surface area (BSA). Your child’s doctor will calculate their BSA using a formula that includes your child’s height in centimeters and weight in kilograms. BSA is measured in meters squared (m2).
Children with a BSA of 1 m2or more typically receive the same dosage as adults: 100 mg twice per day.
The recommended dosage for children with a BSA less than 1 m2 is 100 mg/m2 twice per day.
Dosage questions
Below are answers to some questions you may have about taking Vitrakvi.
What if I miss a dose?
If you miss a dose, try to take it as soon as you remember. But if it’s within 6 hours of your next scheduled dose, you should skip the missed dose. Simply take your next dose at your next scheduled time. You shouldn’t “double up” and take two doses to make up for the missed dose, as this could increase your risk for side effects. (To learn more, see the “Vitrakvi side effects” section below.)
To help make sure that you don’t miss a dose, try using a medication reminder. This can include setting an alarm on your phone or downloading a reminder app. A kitchen timer can work, too.
Will I need to use this drug long term?
Vitrakvi is meant to be used as a long-term treatment. If you and your doctor determine that Vitrakvi is safe and effective for you, you’ll likely take it long term.
Vitrakvi can cause mild or serious side effects. The following lists contain some of the key side effects that may occur while taking Vitrakvi. These lists don’t include all possible side effects.
For more information about the possible side effects of Vitrakvi, talk with your doctor or pharmacist. They can give you tips on how to manage any side effects that may be bothersome.
Note: The Food and Drug Administration (FDA) tracks side effects of drugs it has approved. If you would like to notify the FDA about a side effect you’ve had with Vitrakvi, you can do so through MedWatch.
Mild side effects
Mild side effects* of Vitrakvi can include:
Most of these side effects may go away within a few days or a couple of weeks. But if they become more severe or don’t go away, talk with your doctor or pharmacist.
* This is a partial list of mild side effects from Vitrakvi. To learn about other mild side effects, talk with your doctor or pharmacist, or refer to Vitrakvi’s prescribing information.
† For more information about this side effect, see “Side effect details” below.
Serious side effects
Serious side effects from Vitrakvi aren’t common, but they can occur. Call your doctor right away if you have serious side effects. Call 911 or your local emergency number if your symptoms feel life threatening or if you think you’re having a medical emergency.
Serious side effects of Vitrakvi and their symptoms can include:
- Bone fractures, such as fractures to the hip or leg. Symptoms can include:
- pain
- inability to move the affected area in a typical way
- Allergic reaction.*
- Nervous system problems, such as delirium (sudden and serious changes in behavior and thinking).*
- Liver problems, such as increases in the levels of certain liver enzymes.*
* For more information about this side effect, see “Side effect details” below.
Side effects in children
In clinical studies, Vitrakvi was given to children ages 1 month and older. However, researchers weren’t able to separate data based on age. It’s assumed that side effects of Vitrakvi are the same for children as for adults.
Side effect details
You may wonder how often certain side effects occur with this drug. Here’s some detail on certain side effects this drug may cause.
Allergic reaction
As with most drugs, some people can have an allergic reaction after taking Vitrakvi. But it isn’t known how many people may have had an allergic reaction in clinical trials of the drug.
Symptoms of a mild allergic reaction can include:
- skin rash
- itchiness
- flushing
A more severe allergic reaction is rare but possible. Symptoms of a severe allergic reaction can include:
- swelling under your skin, typically in your eyelids, lips, hands, or feet
- swelling of your tongue, mouth, or throat
- trouble breathing
Call your doctor right away if you have an allergic reaction to Vitrakvi, as the reaction could become severe. Call 911 or your local emergency number if your symptoms feel life threatening or if you think you’re having a medical emergency.
Nervous system problems
Neurological (nervous system) problems are a possible side effect of Vitrakvi. Examples of mild nervous system problems reported by people taking Vitrakvi include:
- confusion
- dizziness
- headache
- problems with coordination
- trouble sleeping
Examples of more serious nervous system problems reported by people taking Vitrakvi include:
- delirium (sudden and serious changes in behavior and thinking)
- encephalopathy (brain disease or damage)
- feelings of tingling, burning, or numbness in the skin
- weakness in the muscles used to speak
- mood disorders
In clinical trials, 42% of people who took Vitrakvi reported having nervous system problems. Vitrakvi wasn’t compared with a different drug or a placebo (treatment with no active drug).
Symptoms of nervous system problems
If you have any of the following symptoms while taking Vitrakvi, talk with your doctor. They could point to a nervous system problem:
- confusion
- trouble talking or speaking
- dizziness
- problems with coordination
- burning, numbness, or tingling in your hands and feet
Depending on how severe your symptoms are, your doctor may lower your dosage or have you stop taking Vitrakvi.
Note: Because of the risk of nervous system problems, you shouldn’t drive a car or operate machinery until you know how Vitrakvi will affect you.
Liver problems
Liver problems were reported in people taking Vitrakvi during clinical trials.
In these trials, 52% of people taking Vitrakvi had an increase in the levels of certain liver enzymes. Researchers found that:
- 6.6% of the increases were considered serious
- 1.1% of people stopped taking Vitrakvi due to the increases
Vitrakvi wasn’t compared with a different drug or a placebo (treatment with no active drug).
While you take Vitrakvi, your doctor will order blood tests to monitor your liver function. They’ll do this every 2 weeks for your first month of treatment. Then your doctor will test your liver function once per month and as needed.
If you have any of the following symptoms while taking Vitrakvi, talk with your doctor. These symptoms could point to a liver problem:
- appetite loss
- nausea or vomiting
- pain in your upper right abdomen (belly)
Depending on how severe your symptoms are, your doctor may lower your dosage or have you stop taking Vitrakvi.
Fatigue
Fatigue was among the more common side effects of Vitrakvi in clinical trials.
Researchers found that:
- 36% of people who took Vitrakvi reported fatigue
- 2.5% of people who took Vitrakvi reported serious fatigue
Vitrakvi wasn’t compared with a different drug or a placebo (treatment with no active drug).
If you have fatigue while taking Vitrakvi that’s bothersome to you, talk with your doctor. They can help determine the best way to ease this side effect.
Nausea and vomiting
Nausea and vomiting are two side effects that were fairly common in Vitrakvi clinical studies.
Researchers found that 25% of people who took the drug reported nausea, and 25% reported vomiting. Vitrakvi wasn’t compared with a different drug or a placebo (treatment with no active drug).
If you experience nausea or vomiting while taking Vitrakvi, you should talk with your doctor. These may be symptoms of liver problems,* which can be another side effect of Vitrakvi. Your doctor will likely check you for other symptoms of liver problems. They may also be able to suggest ways to relieve the nausea or vomiting.
If your nausea or vomiting isn’t related to liver problems, your doctor should still be able to recommend ways to help ease the side effects. But if they become too severe, you may be advised to stop taking Vitrakvi.
* To learn more, see “Liver problems” above.
Cough
Cough is another common side effect that can occur with Vitrakvi use.
In clinical trials, 32% of people taking Vitrakvi experienced a cough. Vitrakvi wasn’t compared with a different drug or a placebo (treatment with no active drug).
While taking Vitrakvi, if you have a cough that’s bothersome to you, talk with your doctor. They can help suggest ways to relieve this side effect.
Vitrakvi is approved to treat solid tumors (cancer) that meet certain conditions:
- have certain genes that have fused together abnormally, and
- are metastatic (have spread) or can’t be removed through surgery because it could cause serious complications, and
- spread after you tried other treatments or other effective treatments aren’t available
For more information about the uses of Vitrakvi, see the “Vitrakvi uses” section above. You can also refer to that section for an explanation of solid tumors and a list of different types of cancers involving solid tumors.
What Vitrakvi does
The active drug in Vitrakvi, larotrectinib, works by blocking the action of enzymes called tropomyosin receptor kinases (TRKs). Enzymes are special proteins that your body uses to help speed up reactions necessary for your body to function.
Certain mutations (changes) in your genes involving TRK enzymes can cause solid tumors. Larotrectinib blocks the action of these changed enzymes to stop your tumor from growing and spreading.
How long does it take to work?
How soon Vitrakvi starts to work for you depends on the size of the tumor and what kind of cancer you have. Your doctor will be able to determine whether the drug is working with blood tests or imaging tests, such as X-rays, CT scans, or MRI scans.
Other drugs are available that can treat solid tumors (cancer). Some may be a better fit for you than others. If you’re interested in finding an alternative to Vitrakvi, talk with your doctor. They can tell you about other medications that may work well for you.
Note: Some of the drugs listed here are used off-label to treat these specific conditions. Off-label drug use is when an FDA-approved drug is used for a purpose other than what it’s approved for.
Alternatives for solid tumors
Examples of other drugs that may be used to treat certain solid tumors anywhere in the body include:
- entrectinib (Rozlytrek)
- pembrolizumab (Keytruda)
- chemotherapy drugs, such as docetaxel and fluorouracil
Examples of other drugs that may be used to treat certain solid tumors in the breast include:
- entrectinib (Rozlytrek)
- pembrolizumab (Keytruda)
- chemotherapy drugs, such as docetaxel (Taxotere) and doxorubicin
- biologic drugs, such as atezolizumab (Tecentriq) and trastuzumab (Herceptin)
- hormonal treatments, such as letrozole (Femara) and anastrozole (Arimidex)
- other medications, such as methotrexate (Trexall)
Examples of other drugs that may be used to treat certain solid tumors in the colon include:
- entrectinib (Rozlytrek)
- pembrolizumab (Keytruda)
- chemotherapy drugs, such as irinotecan or capecitabine (Xeloda)
- biologic drugs, such as bevacizumab (Avastin) or nivolumab (Opdivo)
Examples of other drugs that may be used to treat certain solid tumors in the lung include:
- entrectinib (Rozlytrek)
- pembrolizumab (Keytruda)
- chemotherapy drugs, such as gemcitabine (Gemzar) and doxorubicin
- biologic drugs, such as bevacizumab (Avastin) and durvalumab (Imfinzi)
- targeted therapies, such as afatinib (Gilotrif)
- other medications, such as methotrexate (Trexall)
Examples of other drugs that may be used to treat certain solid tumors in the prostate include:
- entrectinib (Rozlytrek)
- pembrolizumab (Keytruda)
- chemotherapy drugs, such as docetaxel (Taxotere)
- hormonal drugs, such as leuprolide (Lupron Depot) or bicalutamide (Casodex)
- targeted treatments, such as olaparib (Lynparza) or abiraterone (Zytiga)
- other treatments, such as sipuleucel-T (Provenge)
There aren’t any known interactions between taking Vitrakvi and drinking alcohol.
However, both Vitrakvi and alcohol can cause liver problems* including nausea and vomiting. Drinking alcohol while you’re taking Vitrakvi can make these side effects more likely to occur, and it can make them more serious.
Also, according to the
If you have questions about Vitrakvi and alcohol, talk with your doctor.
* To learn more about Vitrakvi and liver problems, see “Liver problems” and “Nausea and vomiting” in the “Vitrakvi side effects” section above.
Vitrakvi can interact with several other medications. It can also interact with certain supplements as well as certain foods.
Different interactions can cause different effects. For instance, some interactions can interfere with how well a drug works. Other interactions can increase the number of side effects or make them more severe.
Vitrakvi and other medications
Below are examples of medications that can interact with Vitrakvi. This section doesn’t contain all drugs that may interact with Vitrakvi.
Before taking Vitrakvi, talk with your doctor and pharmacist. Tell them about all prescription, over-the-counter, and other drugs you take. Also tell them about any vitamins, herbs, and supplements you use. Sharing this information can help you avoid potential interactions.
If you have questions about drug interactions that may affect you, ask your doctor or pharmacist.
Vitrakvi and strong CYP3A4 inhibitors
CYP3A4 is an enzyme that helps your body break down drugs such as Vitrakvi. An enzyme is a type of protein. Certain medications can inhibit (slow down) the activity of CYP3A4. These are called strong CYP3A4 inhibitors.
Taking Vitrakvi with strong CYP3A4 inhibitors can raise the level of Vitrakvi in your body. This could increase the number and severity of side effects from Vitrakvi. (To learn about possible side effects, see the “Vitrakvi side effects” section above.)
Examples of strong CYP3A4 inhibitors include:
- certain antibiotics, such as clarithromycin or erythromycin (Ery-Tab)
- certain antifungals, such as ketoconazole and itraconazole (Sporanox)
- certain protease inhibitors, such as saquinavir (Invirase) or tipranavir (Aptivus)
The manufacturer of Vitrakvi recommends against using a strong CYP3A4 inhibitor with Vitrakvi. However, you may be using a CYP3A4 inhibitor and you and your doctor decide that Vitrakvi is right for you. In this situation, your doctor will likely decrease your Vitrakvi dose. Once you stop using the CYP3A4 inhibitor and it’s out of your system, you’ll likely start taking the regular recommended dose of Vitrakvi.
Before you start using Vitrakvi, tell your doctor about any medications you’re taking. They can determine if any are strong CYP3A4 inhibitors and adjust your treatment plan as needed.
Vitrakvi and strong CYP3A4 inducers
CYP3A4 is an enzyme that helps your body metabolize (break down) drugs. An enzyme is a type of protein. Certain medications can induce (speed up) the activity of CYP3A4. These are called strong CYP3A4 inducers.
Taking Vitrakvi with strong CYP3A4 inducers can lower the level of Vitrakvi in your body. This process could cause Vitrakvi to work less well than usual or not work at all.
Examples of strong CYP3A4 inducers include:
- certain antibiotics, such as rifampin (Rifadin)
- certain anticonvulsants, such as carbamazepine (Tegretol) and phenytoin (Dilantin)
- certain antivirals, such as efavirenz (Sustiva) and nevirapine (Viramune)
- certain corticosteroids, such as dexamethasone (Hemady)
The manufacturer of Vitrakvi recommends against using a strong CYP3A4 inducer with Vitrakvi. However, you may be using a CYP3A4 inducer and you and your doctor decide that Vitrakvi is right for you. In this situation, your doctor will likely increase your Vitrakvi dose. Once you stop using the CYP3A4 inducer and it’s out of your system, you’ll likely start taking the regular recommended dose of Vitrakvi.
Before you start using Vitrakvi, tell your doctor about any medications you’re taking. They can determine if any are strong CYP3A4 inducers and then adjust your treatment plan as needed.
Vitrakvi and herbs and supplements
St John’s wort is a supplement that’s sometimes used to help with mood. Like some of the drugs mentioned above, St. John’s wort is a strong CYP3A4 inducer. The manufacturer of Vitrakvi recommends against using a strong CYP3A4 inducer (such as St. John’s wort) with Vitrakvi.
In some cases, you may be using St. John’s wort and you and your doctor decide that Vitrakvi is right for you. In this situation, your doctor will likely increase your Vitrakvi dose. Once you stop using St. John’s wort and it’s out of your system, you’ll likely start taking the regular recommended dose of Vitrakvi.
Be sure to check with your doctor or pharmacist before using any supplements, including St. John’s wort, while taking Vitrakvi.
Vitrakvi and foods
You should avoid eating grapefruit and drinking grapefruit juice while taking Vitrakvi.
Eating grapefruit or drinking grapefruit juice may increase the level of Vitrakvi in your body. This is because grapefruit products may slow down how quickly your body breaks down the drug. This can increase your risk for side effects or make side effects worse. (To learn more about side effects, see the “Vitrakvi side effects” section above.)
You should take Vitrakvi according to your doctor’s or healthcare professional’s instructions.
Vitrakvi comes in two forms, and you’ll likely be able to take either one.
One form is capsules that you swallow. The other form is a liquid solution. You use an oral syringe to swallow the solution.
If you’re prescribed the liquid solution, a healthcare professional will show you how to use the oral syringes. You should use only these syringes to take Vitrakvi. Your doctor may give you Vitrakvi and the syringes. Or they may have you pick up the drug and oral syringes at a pharmacy.
When to take
Vitrakvi is typically taken twice per day.
To help make sure that you don’t miss a dose, try using a medication reminder. This can include setting an alarm on your phone or downloading a reminder app. A kitchen timer can work, too.
Taking Vitrakvi with food
You can take either Vitrakvi capsules or the liquid solution with or without food.
Can Vitrakvi be crushed, split, or chewed?
No, you shouldn’t crush, split, or chew Vitrakvi capsules. You should swallow them whole.
Based on how Vitrakvi works, the drug may harm the fetus if used during pregnancy.
Some people are born without certain enzymes called tropomyosin receptor kinases (TRKs). Enzymes are special proteins that your body uses to help speed up reactions needed for typical functions. A lack of TRK enzymes has been linked to problems such as:
- developmental delays
- obesity
- anhidrosis (not being able to sweat when you feel hot)
- cognitive impairment, such as thinking more slowly than usual or memory loss
- not being able to feel pain
Vitrakvi works by inhibiting (blocking the action of) TRK enzymes. So it’s thought that taking the drug while pregnant could harm the fetus. As a result, problems such as those mentioned above could occur after birth.
If you’re able to become pregnant, your doctor may give you a pregnancy test before prescribing Vitrakvi. If you are pregnant, your doctor can advise you on the right cancer treatment for you.
If you already know you’re pregnant or plan on becoming pregnant, be sure to tell your doctor before taking Vitrakvi. They can review the risks and benefits of the drug, and work to find the right medication for you.
Vitrakvi and fertility
Clinical trials haven’t studied whether Vitrakvi can affect fertility in humans.
In one animal study, young females given Vitrakvi had decreased fertility. However, other animal studies haven’t shown this effect. And animal studies don’t always predict what will happen in people.
If you have questions about Vitrakvi and fertility, talk with your doctor.
Vitrakvi may cause fetal harm if it’s used during pregnancy. If you’re sexually active and you or your partner can become pregnant, talk with your doctor about your birth control needs while you’re using Vitrakvi.
For more information about taking Vitrakvi during pregnancy, see the “Vitrakvi and pregnancy” section above.
Note: Sex and gender exist on spectrums. Use of the terms “male” and “female” in this article refers to sex assigned at birth.
For females using Vitrakvi
People who are able to become pregnant should use birth control while taking Vitrakvi and for at least 1 week after their last dose.
For males using Vitrakvi
If you’re a male and your partner can become pregnant, you should use birth control while taking Vitrakvi. You should keep using birth control for at least 1 week following your last dose of Vitrakvi.
You shouldn’t breastfeed while taking Vitrakvi and for at least 1 week after your last dose of the drug.
It isn’t known if the drug passes into human breast milk. But based on how Vitrakvi works, the drug may harm a child who is breastfed.
For a list of problems that Vitrakvi may cause in a child who is breastfed, see the “Vitrakvi and pregnancy” section above.
If you’re breastfeeding or plan to breastfeed, talk with your doctor about taking Vitrakvi. They can recommend healthy ways to feed your child and other cancer treatments, if needed.
Here are answers to some frequently asked questions about Vitrakvi.
What types of cancer is Vitrakvi used for?
Vitrakvi is approved to treat certain solid tumors (cancer), which occur with a number of different cancers. These include:
This is not a complete list of the cancers that Vitrakvi is prescribed to treat. You can refer to Vitrakvi’s prescribing information for a full list of the solid tumors the drug is approved to treat. For more information about how Vitrakvi is used, you can also see the “Vitrakvi uses” section above.
Some cancer medications treat tumors depending on where in the body they’re located. This isn’t the case with Vitrakvi. Instead, the tumors must have special genetic markers, which your doctor can look for. The tumors also need to be solid. Vitrakvi isn’t approved to treat cancers of the blood, such as leukemia.
If you have questions about whether Vitrakvi is right for your type of cancer, talk with your doctor.
Will Vitrakvi cure my cancer?
No, Vitrakvi won’t cure your cancer. There isn’t currently a cure for cancer. However, in clinical studies, Vitrakvi has been shown to help slow the growth of solid tumor cancers.
If you have more questions about treatments for your cancer, talk with your doctor.
Can I drive while taking Vitrakvi?
Maybe. It depends on how the drug affects you.
In clinical studies, some people had neurological (nervous system) problems while taking Vitrakvi. Some of the common ones were dizziness and tiredness. Less commonly, Vitrakvi caused side effects such as confusion and problems with coordination.
Because of these possible side effects, you shouldn’t drive a car or operate machinery until you know how Vitrakvi will affect you. If you have more questions about driving while taking Vitrakvi, talk with your doctor.
Will I need to have any tests or monitoring while I take Vitrakvi?
Yes, treatment with Vitrakvi requires some tests and monitoring.
Vitrakvi is approved to treat solid tumors (cancer) with certain abnormal genes. Before you start taking Vitrakvi, your doctor will perform a test to see if your tumor carries these genes. (To learn more about the uses of Vitrakvi, see the “Vitrakvi uses” section above.)
Also, if you’re able to become pregnant, your doctor may give you a pregnancy test before prescribing Vitrakvi. This is because taking the drug while pregnant could harm the fetus. (For more information, see the “Vitrakvi and pregnancy” section above.)
Increases in the levels of certain liver enzymes may occur with Vitrakvi. Because of this, your doctor will likely order blood tests to monitor your liver function. They’ll do this every 2 weeks for your first month of treatment. Then they’ll test your liver function once per month and as needed. (To learn more, see “Liver problems” in the “Vitrakvi side effects” section above.)
If you have more questions on what tests you may need while taking Vitrakvi, talk with your doctor.
How will my doctor decide which form of Vitrakvi to prescribe?
Vitrakvi comes in two forms, and you’ll likely be able to take either one.
One form is capsules that you swallow. The other form is a liquid solution. You’ll use an oral syringe to swallow the solution.
If you’re prescribed the liquid solution, a healthcare professional will show you how to use the oral syringes. You should use only these syringes to take Vitrakvi.
If you have more questions about which form of Vitrakvi is right for you, talk with your doctor.
This drug comes with several precautions. Before taking Vitrakvi, talk with your doctor about your health history. Vitrakvi may not be right for you if you have certain medical conditions or other factors affecting your health. These include:
- Liver problems. One possible side effect of Vitrakvi is liver problems, such as increases in the levels of certain liver enzymes. It isn’t known if this side effect is more likely to occur in people who already have problems. Before taking Vitrakvi, be sure to talk with your doctor about any liver problems you may have.
- Increased risk of bone fractures. Vitrakvi may lead to bone fractures. If you have a condition that increases your risk of fractures, such as osteoporosis, this may make fractures with Vitrakvi more likely. If you experience symptoms of bone fracture during Vitrakvi treatment, tell your doctor. Symptoms may include pain and changes to mobility.
- Nervous system problems. A possible side effect of Vitrakvi is nervous system problems, such as delirium. It isn’t known if this side effect is more likely to occur in people who already have nervous system problems. But before taking Vitrakvi, you should tell your doctor about any nervous system problems you may have. (To learn more, see “Nervous system problems” in the “Vitrakvi side effects” section above.)
- Allergic reaction. If you’ve had an allergic reaction to Vitrakvi or any of its ingredients, you shouldn’t take Vitrakvi. Ask your doctor what other medications are better options for you.
- Pregnancy. Using Vitrakvi while pregnant could harm the fetus. For more information, see the “Vitrakvi and pregnancy” section above.
- Breastfeeding. You shouldn’t breastfeed while taking Vitrakvi. For more information, see the “Vitrakvi and breastfeeding” section above.
Note: For more information about the potential negative effects of Vitrakvi, see the “Vitrakvi side effects” section above.
Do not use more Vitrakvi than your doctor recommends. For some drugs, doing so may lead to unwanted side effects or overdose.
What to do in case you take too much Vitrakvi
If you think you’ve taken too much of this drug, call your doctor. You can also call America’s Poison Centers at 800-222-1222 or use its online tool. But if your symptoms are severe, call 911 or your local emergency number, or go to the nearest emergency room right away.
What if I vomit after taking a dose?
If you vomit after taking your Vitrakvi dose, you shouldn’t take another dose. Simply wait until your next scheduled dose. Taking another dose could increase your risk for side effects or overdose, even if you vomited your first dose.
When you get Vitrakvi from the pharmacy, the pharmacist will add an expiration date to the label on the bottle. This date is typically 1 year from the date they dispensed the medication.
The expiration date helps guarantee that the medication is effective during this time. The
Storage
How long a medication remains good can depend on many factors, including how and where you store the medication.
You should store Vitrakvi capsules at room temperature (68°F to 77°F/20°C to 25°C) in a tightly sealed container away from light. Avoid storing this medication in areas where it could get damp or wet, such as bathrooms.
You should store bottles of Vitrakvi liquid solution in the refrigerator from 36°F to 46°F (2°C to 8°C) until it’s time to take your dose. Don’t freeze Vitrakvi solution.
Disposal
If you no longer need to take Vitrakvi and have leftover medication, it’s important to dispose of it safely. This helps prevent others, including children and pets, from taking the drug by accident. It also helps keep the drug from harming the environment.
Keep in mind that if you have leftover Vitrakvi liquid solution after 90 days of opening the bottle, you should dispose of it.
This article provides several useful tips on medication disposal. You can also ask your pharmacist for information about how to dispose of your medication.
Disclaimer: Medical News Today has made every effort to make certain that all information is factually correct, comprehensive, and up to date. However, this article should not be used as a substitute for the knowledge and expertise of a licensed healthcare professional. You should always consult your doctor or another healthcare professional before taking any medication. The drug information contained herein is subject to change and is not intended to cover all possible uses, directions, precautions, warnings, drug interactions, allergic reactions, or adverse effects. The absence of warnings or other information for a given drug does not indicate that the drug or drug combination is safe, effective, or appropriate for all patients or all specific uses.