Ebola virus disease is a serious, often fatal condition in humans and nonhuman primates. Ebola is one of several viral hemorrhagic fevers, caused by infection with a virus of the Filoviridae family, genus Ebolavirus.
The fatality rates of Ebola vary depending on the strain. For example, Ebola-Zaire can have a fatality rate of up to 90 percent while Ebola-Reston has never caused a fatality in humans.
The infection is transmitted by direct contact with the blood, body fluids, and tissues of infected animals or people. Severely ill patients require intensive supportive care. Ebola virus disease (EVD) is often characterized by the abrupt onset of fever, intense weakness, muscle pain, headache, and sore throat.
Ebola tends to spread quickly through families and friends as they are exposed to infectious secretions when caring for an ill individual. The time interval from infection with Ebola to the onset of symptoms ranges from 2-21 days.
Fast facts on Ebola:
- Ebola is considered a zoonotic virus, meaning that it originated in animals and then spread to humans.
- There is currently no vaccine available for Ebola, although several are in development.
- One vaccine, called Ebola ça suffit, was found to be 100 percent effective in a trial involving 4,000 people in Guinea.
The time interval from infection with Ebola to the onset of symptoms is 2-21 days, although 8-10 days is most common. Signs and symptoms include:
- fever
- headache
- joint and muscle aches
- weakness
- diarrhea
- vomiting
- stomach pain
- lack of appetite
Some patients may experience:
- rash
- red eyes
- hiccups
- cough
- sore throat
- chest pain
- difficulty breathing
- difficulty swallowing
- bleeding inside and outside of the body
Laboratory tests may show low white blood cell and platelet counts and elevated liver enzymes. As long as the patient’s blood and secretions contain the virus, they are infectious. In fact, Ebola virus was isolated from the semen of an infected man 61 days after the onset of illness.
There is currently no licensed vaccine available for Ebola. Several vaccines are being tested, but at this time, none are available for clinical use.
At the moment, treatment for Ebola is limited to intensive supportive care and includes:
- balancing the patient’s fluids and electrolytes
- maintaining their oxygen status and blood pressure
- treating a patient for any complicating infections
Ebola vaccines
In October 2014, the World Health Organization (WHO) organized an expert consultation to assess, test, and eventually license two promising Ebola vaccines:
- cAd3-ZEBOV – GlaxoSmithKline has developed this vaccine in collaboration with the United States National Institute of Allergy and Infectious Diseases (NIH). It uses a chimpanzee-derived adenovirus vector with an Ebola virus gene inserted.
- rVSV-ZEBOV – this was developed by the Public Health Agency of Canada in Winnipeg with NewLink Genetics, a company, located in Ames, IA. The vaccine uses a weakened virus found in livestock; one of its genes has been replaced by an Ebola virus gene.
On July 31 2015,
The next step is to make these vaccines available as soon as possible – and in sufficient quantities – to protect critical frontline workers and to make a difference in the epidemic’s future evolution.
It is still unknown how individuals are infected with Ebola, so stopping infection is still difficult. Preventing transmission is achieved by:
- ensuring all healthcare workers wear protective clothing
- implementing infection-control measures, such as complete equipment sterilization and routine use of disinfectant
- isolation of Ebola patients from contact with unprotected persons
Thorough sterilization and proper disposal of needles in hospitals are essential in preventing further infection and halting the spread of an outbreak.
Ebola tends to spread quickly through families and among friends as they are exposed to infectious secretions when caring for an ill individual. The virus can also spread quickly within healthcare settings for the same reason, highlighting the importance of wearing appropriate protective equipment, such as masks, gowns, and gloves.
Together with the WHO, the Centers for Disease Control and Prevention (CDC) has developed a set of guidelines to help prevent and control the spread of Ebola –
Ebola is caused by viruses in the Ebolavirus and Filoviridae family. Ebola is considered a zoonosis, meaning that the virus is present in animals and is transmitted to humans.
How this transmission occurs at the onset of an outbreak in humans is unknown.
In Africa, people have developed Ebola after handling infected animals found ill or dead, including chimpanzees, gorillas, fruit bats, monkeys, forest antelope, and porcupines.
Person-to-person transmission occurs after someone infected with Ebolavirus becomes symptomatic. As it can take between 2 and 21 days for symptoms to develop, a person with Ebola may have been in contact with hundreds of people, which is why an outbreak can be hard to control and may spread rapidly.
How does Ebola transmission occur in humans?
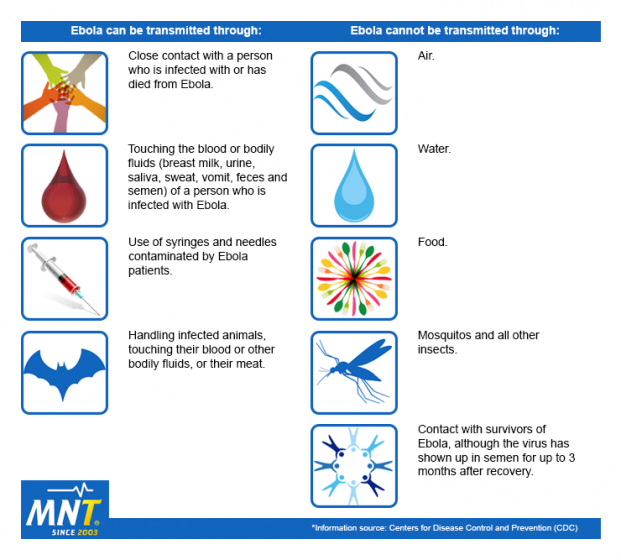
When an Ebola infection occurs in humans, the virus can be spread in several ways to others. Above is a list of ways Ebola can and cannot be transmitted.
Transmission of Ebola between humans can occur through:
- Direct contact through broken skin and mucous membranes with the blood, secretions, organs, or other body fluids of infected people.
- Indirect contact with environments contaminated with such fluids.
- Exposure to contaminated objects, such as needles.
- Burial ceremonies in which mourners have direct contact with the body of the deceased.
- Exposure to the semen of people with Ebola or who have recovered from the disease – the virus can still be transmitted through semen for up to 7 weeks after recovery from illness.
- Contact with patients with suspected or confirmed EVD – healthcare workers have frequently been infected while treating patients.
There is no evidence that Ebola can be spread via insect bites.
The first cases of Ebola were reported simultaneously in 1976 in Yambuku, near the Ebola River in Zaire (now the Democratic Republic of the Congo) and in Nzara, Sudan.
Since then, eruptions or asymptomatic cases of Ebola in humans and animals have surfaced intermittently in the following locations due to outbreaks, laboratory contamination, and accidents:
- The Democratic Republic of the Congo (DRC)
- Sudan (South Sudan)
- Senegal
- United Kingdom
- United States (U.S.)
- Philippines
- Italy
- Spain
- Gabon
- Ivory Coast
- South Africa
- Russia
- Uganda
- Guinea
- Liberia
- Sierra Leone
The 2014 Ebola outbreak was the largest in history, primarily affecting Guinea, northern Liberia, and Sierra Leone. The Centers for Disease Control and Prevention (CDC) estimates that the epidemic caused more than 11,000 deaths, with almost all occurring in West Africa.
In the U.S., reports indicate that there have been two imported cases, including one death, and two locally acquired cases in healthcare workers.
A small number of cases were reported in Nigeria, Mali, and Senegal, with health authorities able to contain these cases and prevent further spread.
The risk of contracting Ebola is low. There is a higher risk of becoming infected when:
- Traveling to areas of Africa where there have been confirmed cases of Ebola.
- Conducting animal research with monkeys imported from Africa or the Philippines.
- Providing medical or personal care to people who may have been exposed to Ebola.
- Preparing people for burial who have been infected with Ebola.
Tests and diagnosis
According to the WHO, samples from patients with Ebola are an extreme biohazard risk. Testing should be conducted under maximum biological containment conditions.
Before Ebola can be diagnosed, other diseases should be ruled out, and, if Ebola is suspected, the patient should be isolated. Public health professionals should be notified immediately. Ebola virus infections can be diagnosed definitively in a laboratory through several types of tests, including:
- Antigen-capture enzyme-linked immunosorbent assay (ELISA) testing.
- IgM ELISA.
- Polymerase chain reaction (PCR).
- Virus isolation.
In the more advanced stages of the disease or after recovery, diagnosis is made using IgM and IgG antibodies. Ebola can be diagnosed retrospectively in deceased patients by other forms of testing.
