Humira is a brand-name prescription medication. It’s FDA-approved to treat several conditions, including:
- rheumatoid arthritis (RA) in adults
- juvenile idiopathic arthritis (JIA) in children ages 2 years and older
- psoriatic arthritis (PsA) in adults
- ankylosing spondylitis (AS) in adults
- Crohn’s disease (CD) in adults as well as children ages 6 years and older
- ulcerative colitis (UC) in adults as well as children ages 5 years and older
- plaque psoriasis in adults
- hidradenitis suppurativa (HS) in adults as well as children ages 12 years and older
- certain types of uveitis in adults as well as children ages 2 years and older
Humira has limitations to its use in treating UC. For details on how Humira is used to treat all of these conditions, see the sections “Humira for plaque psoriasis” and “Other uses for Humira” below.
Humira drug class and form
Humira contains the active drug adalimumab, which is a biologic medication. Biologics are medications made from living cells. Adalimumab belongs to a class of medications known as tumor necrosis factor (TNF) blockers. (A class of medications is a group of drugs that work in a similar way.) TNF is a protein made by your body.
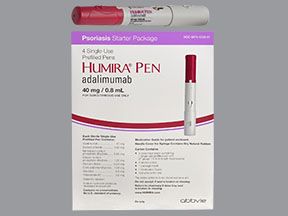
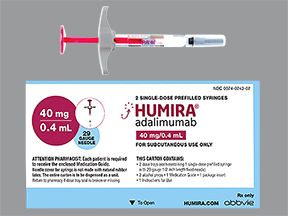
Humira comes in three forms: a single-dose pen, a single-dose prefilled syringe, and a single-dose vial of liquid solution. To learn the strengths of the forms, see the “Humira dosage” section below.
The drug is given as a subcutaneous injection. A healthcare professional can give you Humira injections. But you may be able to give yourself injections at home with the pen or syringe if your doctor approves. You’d need to be trained first. The vial form of Humira can be used only by healthcare professionals. You won’t be able to use vials yourself.
Effectiveness
In clinical studies, Humira was effective in treating the conditions listed above. For information on some of the results of these studies, see the sections “Humira for plaque psoriasis” and “Other uses for Humira” below.
Humira can cause mild or serious side effects. The following lists contain some of the key side effects that may occur while taking Humira. These lists don’t include all possible side effects.
For more information on the possible side effects of Humira, talk with your doctor or pharmacist. They can give you tips on how to deal with any side effects that may be bothersome.
Note: The Food and Drug Administration (FDA) tracks side effects of drugs they have approved. If you would like to report to the FDA a side effect you’ve had with Humira, you can do so through MedWatch.
Mild side effects
Mild side effects of Humira can include:*
- injection site reactions (itching, pain, or swelling near the injection spot)
- upper respiratory infections, such as the common cold
- headache
- rash
Most of these side effects may go away within a few days or a couple of weeks. But if they become more severe or don’t go away, talk with your doctor or pharmacist.
* This is a partial list of mild side effects from Humira. To learn about other mild side effects, talk with your doctor or pharmacist, or visit Humira’s prescribing information.
Serious side effects
Call your doctor right away if you have serious side effects. Call 911 if your symptoms feel life threatening or if you think you’re having a medical emergency.
Serious side effects and their symptoms can include:
- Heart failure. Symptoms can include:
- feeling short of breath
- sudden weight gain
- swelling in your ankles or feet
- Lupus-like syndrome (an immune system reaction). Symptoms can include:
- chest discomfort or pain
- feeling short of breath
- joint pain
- rash on your arms or cheeks that gets worse in the sun
- Nerve disorders or demyelinating diseases, such as seizures or multiple sclerosis (MS). Symptoms can include:
- dizziness
- numbness or tingling
- weakness in your arms or legs
- vision problems
- Blood disorders, such as anemia (low level of red blood cells). Symptoms can include:
- bruising
- bleeding
- fever that lasts
- unusual pale appearance to the skin
- Liver damage. Symptoms can include:
- loss of appetite
- belly pain
- vomiting
- jaundice (yellow coloring of the skin and white of the eyes)
Other serious side effects, explained in more detail below in “Side effect details,” include:
- allergic reaction
- risk of cancer,* such as lymphoma (cancer of the white blood cells)
- risk of serious infections,* such as tuberculosis (TB) or pneumonia
* Humira has
Side effects in children
In clinical studies, side effects in children who received Humira were similar to those seen in adults, regardless of the condition Humira was used to treat.
Side effect details
You may wonder how often certain side effects occur with this drug. Here’s some detail on some of the side effects this drug may cause.
Allergic reaction
As with most drugs, some people can have an allergic reaction after taking Humira. In a clinical study of children with juvenile idiopathic arthritis (JIA), 6% of those who were given Humira had a mild allergic reaction during the first 48 weeks of treatment. It’s not known how many children who took a placebo (treatment with no active drug) had a mild allergic reaction.
And in a clinical study of children with Crohn’s disease (CD), 5% of those who were given Humira had a mild allergic reaction. It’s not known how many children who took a placebo had a mild allergic reaction.
It’s also not known how often allergic reactions occurred in people who took Humira for the other conditions that the drug treats.
Symptoms
Symptoms of a mild allergic reaction can include:
- skin rash
- itchiness
- flushing
A more severe allergic reaction is rare but possible. It’s not known how often this occurred in clinical trials of Humira.
Symptoms of a severe allergic reaction can include:
- swelling under your skin, typically in your eyelids, lips, hands, or feet
- swelling of your tongue, mouth, or throat
- trouble breathing
Call your doctor right away if you have a severe allergic reaction to Humira. Call 911 if your symptoms feel life threatening or if you think you’re having a medical emergency.
Long-term side effects
Humira can cause some long-term side effects. For example, the drug can cause the growth of new cancers or may cause existing tumors (masses of cancerous tissue) to grow. For more information, see the “Cancer, such as lymphoma” section below.
If you have any concerns about long-term side effects of your Humira treatment, talk with your doctor.
Injection site reaction
An injection site reaction is common while taking Humira. In clinical studies, this was the most commonly reported side effect. About 20% of people who took Humira reported injection site reactions, compared with 14% who took a placebo.
Most reactions are mild and aren’t a reason to stop taking the drug unless they’re having an impact on your daily life.
Examples of injection site reactions include swelling, redness or deepening of skin color, itching, pain, or bleeding where you had the injection.
If you have an injection site reaction during your Humira treatment, tell your doctor. They may be able to suggest ways to ease your discomfort and help with future injections.
Weight gain
Weight gain wasn’t reported as a side effect in clinical studies of Humira. However, weight gain has been reported with the use of other medications called tumor necrosis factor (TNF) blockers. Humira is a type of TNF blocker. Also, weight gain could be a symptom of heart failure, which is another possible side effect of Humira.
If you notice an increase in your weight while you’re using Humira, contact your doctor.
Psoriasis study
A
There were 178 courses of treatments. In 54 treatments with adalimumab, people gained an average of about 5 pounds (lb), or 2.4 kilograms (kg).* In comparison, in the 63 treatments with infliximab, people gained an average of about 3 lb (1.3 kg). People who took other TNF blockers didn’t see significant weight gain over the 48 weeks.
But it isn’t clear whether the weight gain was due to the drug itself, lifestyle factors, or a mix of both.
* One kg equals about 2.2 lb.
Rheumatoid arthritis studies
One study looked at people with rheumatoid arthritis (RA) who took the TNF blockers etanercept or adalimumab or the drug methotrexate (Trexall) over 24 months. The researchers wanted to see how etanercept and adalimumab compared with methotrexate in regard to weight gain. The researchers found that people who took etanercept or adalimumab were six times more likely to gain weight than people who took methotrexate.
Another study looked at 168 people with RA who took a TNF blocker over 24 months. Researchers wanted to see if these people gained weight during treatment. By the end of the study, the average weight gain was about 4 lb (1.8 kg), and 64.3% of people who took a TNF blocker had gained weight. The TNF blocker wasn’t compared with a different drug or a placebo.
It isn’t known for certain whether Humira itself causes weight gain or how much is from diet and lifestyle factors. If you’re concerned about weight gain while taking Humira, talk with your doctor. They can help review your diet and exercise routine.
Cancer, such as lymphoma
Humira has a
Study results
Clinical studies looked at adults who took Humira for any of the conditions the drug is approved to treat. Researchers found that these people were three times more likely to develop lymphoma than people in the United States who didn’t take the drug.
A rare form of lymphoma called hepatosplenic T-cell lymphoma has been reported in children and young adult males.* Most of these cases were in males with Crohn’s disease (CD) or ulcerative colitis (UC).
Besides lymphoma, the second most commonly observed cancer was a type of skin cancer called nonmelanoma. This was followed by breast cancer, colon cancer, prostate cancer, and lung cancer.
It’s not known whether Humira really causes cancer. A review of clinical studies found that there was conflicting evidence on whether TNF blockers increased cancer risk. Analyses of studies and registries that gather information from larger groups also reported conflicting results.
It’s important to note that some of the conditions Humira is used to treat, including RA, are linked to higher risks of developing lymphoma.
* Sex and gender exist on spectrums. Use of the term “male” in this article refers to sex assigned at birth.
Symptoms of lymphoma
Symptoms of lymphoma can include:
- swollen lymph nodes
- chills
- weight loss
- fatigue (lack of energy)
- bruising easily or bleeding easily
If you’re concerned about developing cancer while taking Humira, talk with your doctor. They can help determine the risks and benefits of taking the drug. They may also recommend different medications.
Hair loss
Hair loss wasn’t reported as a side effect during clinical studies of Humira. However, since the drug was released onto the market, there have been reports linking Humira to hair loss.
One review found 62 cases of different types of hair loss in people who had used a TNF blocker. (Humira is a TNF blocker.) People with hair loss usually have a few small patches of hair loss and often recover after stopping the use of the TNF blocker.
Because these reports came out after the drug’s clinical studies, at this time it isn’t possible to say what role Humira played in causing hair loss.
If you’re concerned about hair loss while taking Humira, talk with your doctor.
Skin rash
A skin rash may occur while taking Humira. This is one of the most common side effects of the drug. In clinical studies of people with RA, 12% of people who took Humira reported a rash, compared with 6% of people who took a placebo. The rates of skin rash were similar in people who took Humira for the other conditions that the drug treats.
Most of these cases were mild and went away on their own. Only 0.3% of people across all studies stopped taking Humira due to rash.
If you develop a rash while taking Humira, tell your doctor. They may be able to recommend treatments to help you feel more comfortable.
It’s important to note that a rash can also be a sign of an allergic reaction to Humira. Call your doctor right away if you develop symptoms of a severe allergic reaction to the drug. (See the “Allergic reaction” section above for symptoms.) And call 911 if your symptoms feel life threatening or if you think you’re having a medical emergency.
Infections
Humira has a
Study results
In clinical studies of all approved uses of Humira, people had serious infections caused by bacteria, fungi, and viruses. People who took Humira had higher rates of infection than people who took a placebo. Some of these infections were severe enough to require a hospital stay, and some were fatal.
In the Humira group, there were 4.3 serious infections for every 100 patient years. This means that if 100 people took Humira for a year, 4.3 serious infections would occur. In the placebo group, there were 2.9 serious infections for every 100 patient years.
Before you start to take Humira, your doctor may test you for TB. If your doctor believes that you’re at risk for TB or have an existing TB infection, they may treat you for TB before and during treatment with Humira. They may also test you for TB while you take the drug. This is because you can still develop a TB infection while using Humira.
Symptoms
Symptoms of a serious infection, such as TB, may include:
- coughing up blood
- fever
- cough that lasts
- unexplained weight loss
If you notice any of these symptoms while taking Humira, tell your doctor right away. They’ll likely pause your Humira treatment to treat the infection.
As with all medications, the cost of Humira can vary. There may be additional costs for Humira injections if you receive the injection at your doctor’s office rather than giving them to yourself. In any case, the actual price you’ll pay depends on your insurance plan, your location, and the pharmacy you use.
It’s important to note that you may have to get Humira at a specialty pharmacy. This type of pharmacy is authorized to carry specialty medications. These are drugs that may be expensive or may require help from healthcare professionals to be used safely and effectively.
Your insurance plan may require you to get prior authorization before approving coverage for Humira. This means that your doctor and insurance company will need to communicate about your prescription before the insurance company will cover the drug. The insurance company will review the request and let you and your doctor know if your plan will cover Humira.
If you’re not sure if you’ll need to get prior authorization for Humira, contact your insurance company.
Financial and insurance assistance
If you need financial support to pay for Humira, or if you need help understanding your insurance coverage, help is available.
AbbVie, Inc., the manufacturer of Humira, offers a savings card to help lower the cost of Humira. The Humira Complete Savings Card can be used by people with commercial insurance.
The manufacturer also offers patient assistance programs to help lower Humira’s cost without insurance. And there is support available for people with Medicare coverage.
For more information about Humira Complete resources and to find out if you’re eligible for support, call 800-4HUMIRA (800-448-6472) or visit the program website.
Biosimilar version
The Food and Drug Administration (FDA) has approved seven biosimilar versions of Humira: Hyrimoz, Hadlima, Amjevita, Cyltezo, Abrilada, Hulio, and Yusimry. These biosimilars are not yet available for use.
A biosimilar is a medication that’s similar to a brand-name drug. A generic medication, on the other hand, is an exact copy of a brand-name drug. Biosimilars are based on biologic medications, which are created from parts of living organisms. Generics are based on regular drugs made from chemicals. Biosimilars and generics also tend to cost less than brand-name medications.
The Humira dosage your doctor prescribes will depend on several factors. These include:
- the type and severity of the condition you’re using Humira to treat
- your age
- the form of Humira you take
- other medical conditions you may have
- body weight, in children
Typically, your doctor will start you on a low dosage. Then they’ll adjust it over time to reach the amount that’s right for you. Your doctor will ultimately prescribe the smallest dosage that provides the desired effect.
The following information describes dosages that are commonly used or recommended. However, be sure to take the dosage your doctor prescribes for you. Your doctor will determine the best dosage to fit your needs.
Drug forms and strengths
Humira comes in three forms:
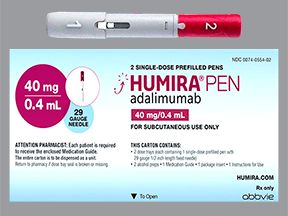
- A single-dose pen. It’s available in these strengths:
- 40 milligrams (mg)/0.4 milliliter (mL)
- 40 mg/0.8 mL
- 80 mg/0.8 mL
- A single-dose prefilled syringe. It’s available in these strengths:
- 10 mg/0.1 mL
- 10 mg/0.2 mL
- 20 mg/0.2 mL
- 20 mg/0.4 mL
- 40 mg/0.4 mL
- 40 mg/0.8 mL
- 80 mg/0.8 mL
- A single-dose vial of liquid solution. It’s available in one strength: 40 mg/0.8 mL.
For more information on the drug’s forms and how they’re given, see the “Humira pen and other forms” section above.
Dosage for adults
The dosages of Humira for adults may vary depending on what condition the drug is treating.
Dosage for plaque psoriasis
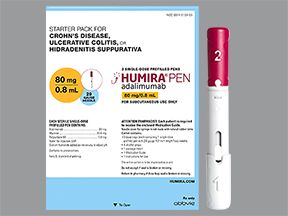
For plaque psoriasis, the recommended starting dose of Humira is 80 mg. This is followed by a dosage of 40 mg every other week, starting the week after you have your first injection.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct pen dosage during your first month of taking Humira.
Dosage for hidradenitis suppurativa (HS)
For hidradenitis suppurativa, the recommended dosage of Humira is as follows:
- You’ll first have a loading dose of 160 mg. A loading dose is designed to quickly get the drug into your body so it can start working right away. You might receive the loading dose as one dose in 1 day or split over 2 back-to-back days.
- Two weeks later (day 15), you’ll have a dose of 80 mg.
- Two weeks later (day 29), you’ll start having a 40-mg injection each week or 80 mg every other week.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct pen dosage during your first month of taking Humira.
Dosage for rheumatoid arthritis (RA)
For rheumatoid arthritis, the recommended dosage of Humira is 40 mg every other week.
If you’re not taking methotrexate (Trexall) with Humira for RA, your doctor may increase your dosage to 40 mg every week or 80 mg every other week.
Dosage for psoriatic arthritis (PsA)
For psoriatic arthritis, the recommended dosage of Humira is 40 mg every other week.
Dosage for ulcerative colitis (UC)
For ulcerative colitis, the recommended dosage of Humira is as follows:
- You’ll first have a loading dose of 160 mg. You might receive this as one dose in 1 day or split over 2 back-to-back days.
- Two weeks later (day 15), you’ll have a dose of 80 mg.
- Two weeks later (day 29), you’ll start having a 40-mg injection every other week.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct pen dosage during your first month of taking Humira.
Dosage for Crohn’s disease (CD)
For Crohn’s disease (CD), the recommended dosage of Humira is as follows:
- You’ll first have a loading dose of 160 mg. You might receive this as one dose in 1 day or split over 2 back-to-back days.
- Two weeks later (day 15), you’ll have a dose of 80 mg.
- Two weeks later (day 29), you’ll start having a 40-mg injection every other week.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct pen dosage during your first month of taking Humira.
Dosage for uveitis
For uveitis, the recommended starting dose of Humira is 80 mg. This is followed by a dosage of 40 mg every other week, starting the week after you have your first injection.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct pen dosage during your first month of taking Humira.
Dosage for ankylosing spondylitis (AS)
For ankylosing spondylitis, the recommended dosage of Humira is 40 mg every other week.
Pediatric dosage
Here’s some information on the Humira dosages in children. Children’s dosing is based on the child’s body weight in kilograms (kg). One kg equals about 2.2 pounds (lb).
Pediatric dosage for hidradenitis suppurativa (HS)
When used for HS in children ages 12 years and older, Humira is dosed based on body weight as follows:
- Children weighing 30 kg to less than 60 kg (about 66 lb to 131 lb): On day 1, your child will receive an80-mg loading dose. On day 8, they’ll receive 40 mg. After that, they’ll receive 40 mg every other week.
- Children weighing 60 kg (about 132 lb) or more: Your child will receivea 160-mg loading dose on day 1, or it can be split into two doses to be given on 2 back-to-back days. On day 15, they’ll receive 80 mg. Then, starting on day 29, they’ll receive either 40 mg every week or 80 mg every other week.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct pen dosage during your child’s first month of taking Humira.
Pediatric dosage for ulcerative colitis (UC)
When used for UC in children ages 5 years and older, Humira is dosed based on body weight as follows:
- Children weighing 20 kg to less than 40 kg (about 44 lb to 87 lb): On day 1, your child will receive an 80-mg loading dose. On day 8, they’ll receive a 40-mg dose. And on day 15, they’ll receive another 40-mg dose. Then starting on day 29 of treatment, they’ll receive either 40 mg every other week or 20 mg each week.
- Children weighing 40 kg (about 88 lb) or more: Your child will receivea 160-mg loading dose on day 1, or this can be split into two doses given on 2 back-to-back days. On day 8, they’ll receive an 80-mg dose. And on day 15, they’ll receive another 80-mg dose. Then starting on day 29, they’ll receive either 80 mg every other week or 40 mg each week.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct pen dosage during your child’s first month of taking Humira.
Pediatric dosage for Crohn’s disease (CD)
When used for Crohn’s disease in children ages 6 years and older, Humira is dosed based on body weight as follows:
- Children weighing 17 kg to less than 40 kg (about 37 lb to 87 lb): On day 1, your child will receive an 80-mg loading dose. On day 15, they’ll receive 40 mg. Then starting on day 29, they’ll receive 20 mg every other week.
- Children weighing 40 kg (about 88 lb) or more: Your child will receive a 160-mg loading dose on day 1, or it can be split into two doses to be given on 2 back-to-back days. On day 15, they’ll receive 80 mg. On day 29, they’ll receive 40 mg. After that, they’ll receive 40 mg every other week.
To help you follow the dosing schedule described above, Humira comes in a starter kit that contains several prefilled pens or syringes. This kit helps make sure you use the correct dosage during your child’s first month of taking Humira.
Pediatric dosage for uveitis
When used for uveitis in children ages 2 years and older, Humira is dosed based on body weight as follows:
- Children weighing 10 kg to less than 15 kg (about 22 lb to 32 lb): 10 mg every other week
- Children weighing 15 kg to less than 30 kg (about 33 lb to 65 lb): 20 mg every other week
- Children weighing 30 kg (about 66 lb) or more: 40 mg every other week
Pediatric dosage for juvenile idiopathic arthritis (JIA)
For juvenile idiopathic arthritis (JIA) in children ages 2 years and older, Humira dosages are based on body weight:
- Children weighing 10 kg to less than 15 kg (about 22 lb to 32 lb): 10 mg every other week
- Children weighing 15 kg to less than 30 kg (about 33 lb to 65 lb): 20 mg every other week
- Children weighing 30 kg (about 66 lb) or more: 40 mg every other week
What if I miss a dose?
If you forget a dose of Humira, take your next dose as soon as you remember. Then, you should take your next dose at the regular time to put you back on schedule. If you receive Humira at your doctor’s office, call the office right away to reschedule your missed dose.
If you aren’t sure what to do after missing a dose of Humira, talk with your doctor.
To help make sure that you don’t miss a dose, try using a medication reminder. This can include setting an alarm or using a timer. You could also download a reminder app on your phone.
Will I need to use this drug long term?
Humira is meant to be used as a long-term treatment. If you and your doctor determine that Humira is safe and effective for you, you’ll likely take it long term.
Humira is a brand-name medication. The Food and Drug Administration (FDA) has approved seven biosimilar versions of Humira: Hyrimoz, Hadlima, Amjevita, Cyltezo, Abrilada, Hulio, and Yusimry. However, these biosimilars are not yet available for use.
A biosimilar is a medication that’s similar to a brand-name drug. A generic medication, on the other hand, is an exact copy of a brand-name drug. Biosimilars are based on biologic medications, which are created from parts of living organisms. Generics are based on regular drugs made from chemicals. Biosimilars and generics also tend to cost less than brand-name medications.
Humira contains the active drug adalimumab. This means adalimumab is the ingredient that makes Humira work.
The Food and Drug Administration (FDA) approves prescription drugs such as Humira to treat certain conditions. Humira is FDA-approved to treat moderate to severe plaque psoriasis in adults that’s chronic (long term).
Plaque psoriasis is a condition in which the immune system mistakenly attacks the body, causing inflammation (swelling). As a result, plaques (patches) form on the skin and may be covered by silvery scales. The plaques are often painful and itchy. Symptoms of plaque psoriasis tend to worsen during times called flare-ups and improve during periods known as remission.
To use Humira, you must be eligible for phototherapy or systemic therapy. Phototherapy uses light to treat plaque psoriasis. And systemic therapy refers to medications that work through your entire body to help stop plaques from forming. Humira is meant for people with plaque psoriasis that would benefit more from Humira than it would from other systemic therapies. The drug is also for people who can’t use other systemic therapies.
In addition, Humira is approved for other conditions, which are discussed in the “Other uses for Humira” section below.
Humira treats plaque psoriasis by blocking a protein called tumor necrosis factor (TNF). This decreases the activity of your immune system, leading to a reduction in the number and severity of skin plaques.
Effectiveness for plaque psoriasis
Clinical studies have shown that Humira is effective for treating plaque psoriasis. These studies looked at adults with moderate to severe plaque psoriasis.
In one study, Humira was compared with a placebo (treatment with no active drug). Among people who received 40 mg of Humira every other week, 71% reported that their psoriasis symptoms eased by at least 75%. This is in comparison with the placebo group, in which only 7% of people reported that their symptoms eased by 75% or more.
The Food and Drug Administration (FDA) approves prescription drugs such as Humira to treat certain conditions. In addition to treating plaque psoriasis, which is presented in the section just above, Humira is also approved for other uses. These other uses are described below.
Humira for hidradenitis suppurativa (HS)
Humira is FDA-approved to treat moderate to severe hidradenitis suppurativa, a chronic (long-term) skin condition. The main symptoms are painful skin breakouts that can occur in areas such as the armpits, groin, and inner thighs. These breakouts are often painful and can take numerous forms, including pimple-like red bumps, cysts or nodules, and boils. The breakouts can be triggered or worsened by things like stress, heat, and hormonal changes.
It’s not known exactly what leads to HS, but an overactive immune system is believed to be one possible cause. Humira treats HS by blocking a protein called tumor necrosis factor (TNF). This decreases the activity of your immune system, leading to a reduction in the number and severity of skin breakouts.
Effectiveness for hidradenitis suppurativa
Humira has been proven effective in adults with moderate to severe HS.
In clinical studies, people with HS received either Humira or a placebo. People in both groups also used a topical antiseptic wash each day. (This is a wash applied to the skin that helps fight germs.) Researchers wanted to see if Humira was better than a placebo at reducing the number of skin lesions (sores) and breakouts by at least 50%. This is defined as a “clinical response.”
The researchers found that 42% to 59% of people who took Humira had a clinical response, compared with only 26% to 28% of people who took a placebo.
The use of Humira in children ages 12 years and older was based on clinical studies of adults. The safety and effectiveness of the drug is estimated to be the same in children as in adults. Dosing for children ages 12 years and older is based on their weight.
Humira for ulcerative colitis (UC)
Humira is FDA-approved to treat moderate to severe UC that’s active. “Active” means that you have symptoms. Humira can be used for UC in adults as well as children 5 years and older.
Before starting Humira, you must’ve already tried drugs called immunosuppressants, but they didn’t work for you. Examples of these immunosuppressants include 6-mercaptopurine (6-MP), azathioprine, and corticosteroids.
Humira wasn’t studied in people with UC who had previously used other TNF blocker drugs. (Humira is a TNF blocker drug.) It’s not known if Humira is effective in people who couldn’t take TNF blockers or whose condition didn’t improve with them.
The goal of Humira is to help you reach remission, which is a time when your symptoms ease, become less frequent, or disappear.
UC explained
UC is a form of inflammatory bowel disease, which is a type of disease that affects your digestive tract. With UC, you have inflammation (swelling) of the lining of your large intestine (colon), rectum, or both.
The inflammation results in tiny sores, called ulcers, throughout the lining of your colon. This causes your bowels to move their contents rapidly and empty frequently. As a result, you may have symptoms such as diarrhea, belly pain, malnutrition (lack of nutrients), weight loss, and bloody stools.
It’s thought that UC results from an overactive immune system. Humira treats UC by blocking a protein called TNF. This decreases the activity of your immune system, leading to a reduction in the number and severity of colon ulcers.
Effectiveness for ulcerative colitis
Humira was studied in adults with moderate to severe UC that was active. These people had either tried or were currently taking other drugs for UC called immunosuppressants. Some people were also taking drugs known as aminosalicylates.
In clinical studies, researchers wanted to see if Humira was better than a placebo at causing symptoms to go into remission. In one study, 18.5% of people who took Humira had symptom remission after 8 weeks, compared with 9.2% of people who took a placebo.
A similar study found that 16.5% of people who took Humira had symptom remission after 8 weeks, compared with 9.3% of people who took a placebo. At 52 weeks, 8.5% of people who took Humira were still in symptom remission, compared with 4.1% of people who took a placebo.
Study results in children
Humira was studied in children ages 5 years and older with moderate to severe UC that was active. The children had already tried or were currently taking certain other drugs for UC. These drugs included corticosteroids, an immunomodulator, or a TNF blocker.
In clinical studies, researchers looked at whether certain dosages of Humira were better than a placebo at causing remission of UC symptoms. After 8 weeks of treatment, 60% of children who took the recommended dosage of Humira experienced remission. Also, 43% of children who took a lower Humira dosage experienced remission of their symptoms.
After a total of 52 weeks of treatment, 45% of children who took the recommended Humira dosage experienced remission of their UC symptoms. This is compared to 29% of children who took a lower Humira dosage, and 33% of children who took a placebo.
Humira for Crohn’s disease (CD)
Humira is FDA-approved to treat moderate to severe Crohn’s disease (CD) in adults that’s active. “Active” means that you have symptoms.
The goal of Humira is to ease the signs and symptoms of CD and help you reach remission, which is a time when you don’t have symptoms.
CD explained
CD is a form of inflammatory bowel disease. CD can affect any part of your digestive tract, but it most commonly occurs in the small intestine and the large intestine (colon). CD often causes symptoms such as diarrhea, stomach cramps, bloody stools, fatigue (lack of energy), weight loss, and frequent bowel movements.
It’s believed that an overactive immune system is a factor in causing CD. Humira treats CD by blocking a protein called TNF. This decreases the activity of your immune system, leading to a reduction in the number and severity of CD symptoms.
Effectiveness for Crohn’s disease in adults
In adults with CD, Humira has been shown to be effective at causing and maintaining both remission and significantly easing CD symptoms.
In clinical studies, people with CD were treated with either Humira (at various doses) or a placebo. Researchers found that 21% to 36% of people who took Humira had a CD remission by week 4. This is compared with 7% to 12% of people who took a placebo.
Of the people who took Humira, 52% to 58% found that their CD symptoms eased significantly by week 4, compared with 34% of people who took a placebo.
Another study looked at maintaining remission and symptom reduction at weeks 26 and 56. In this study, people took either Humira or a placebo for maintenance of CD. At week 26, 40% of the people who took Humira had a CD remission, compared with only 17% of people who took a placebo. By week 56, 36% of the people who took Humira were still in remission, compared with just 12% of people who took a placebo.
Of the people who took Humira, 54% found that their symptoms eased significantly compared with 28% of people who took a placebo by week 26. By week 56, 43% of the people who took Humira found that their symptoms eased significantly, compared with just 18% of people who took a placebo.
Humira for Crohn’s disease in children
Humira is FDA-approved for use in children ages 6 years and older with moderate to severe Crohn’s disease (CD) that’s active. The goal of Humira is to ease the signs and symptoms of CD and help children reach remission.
In clinical studies, Humira has been shown to be effective for moderate to severe CD in children ages 6 to 17 years. In these studies, all children received Humira. There was no placebo group. The children randomly received either a low or high dose of the drug relative to their weight.
Depending on which dose was used, at week 26, 28% to 39% of children had a remission of CD symptoms. And 48% to 59% had CD symptoms that eased significantly.
At week 52, 23% to 33% of children had a remission of their CD symptoms. And 28% to 42% of children found that their symptoms eased significantly.
Humira for rheumatoid arthritis (RA)
Humira is FDA-approved to treat moderate to severe rheumatoid arthritis that’s active in adults. “Active” means that you have symptoms. The goal of Humira is to significantly ease signs and symptoms of RA and help limit joint damage and improve mobility.
RA is a chronic (long-term) autoimmune disease that causes joint pain and inflammation throughout your body. “Autoimmune” means that your immune system mistakenly attacks your body. With RA, there are often periods of flare-ups, in which symptoms occur or get worse, and periods of remission, times when symptoms disappear.
Humira treats RA by blocking a protein called TNF. This decreases the activity of your immune system, leading to a reduction in the number and severity of RA symptoms.
Effectiveness for rheumatoid arthritis
Humira has been proven effective for treating RA in adults.
Clinical studies of people with RA compared Humira with a placebo. In some studies, people also took methotrexate with either Humira or a placebo. Researchers wanted to see how effective Humira was at helping people achieve a 20%, 50%, or 70% reduction in the severity and number RA symptoms. Across these studies, researchers found that Humira was more effective than the placebo.
In two studies, 12% to 21% of people who took Humira by itself or with methotrexate had a 70% reduction in the severity and number of RA symptoms after 6 months. This was compared with just 2% to 3% of people who took a placebo by itself or with methotrexate.
After 12 months, 23% of people who took Humira with methotrexate had a 70% reduction in the severity and number of RA symptoms. In comparison, 5% of people who took a placebo with methotrexate had a 70% reduction.
Humira for psoriatic arthritis (PsA)
Humira is FDA-approved to treat psoriatic arthritis that’s active in adults. “Active” means that you have symptoms. The goal of Humira is to reduce the signs and symptoms of PsA, help prevent the condition from getting worse, and improve mobility.
PsA is a type of arthritis that can occur in people with psoriasis. PsA results in red, scaly patches on the skin and scalp, plus classic arthritis symptoms of swollen, sore joints. With PsA, there are often periods of flare-ups, in which symptoms occur or get worse, and periods of remission, times when symptoms disappear.
PsA occurs when your immune system mistakenly attacks your joints and skin. Humira treats PsA by blocking a protein called TNF. This decreases the activity of your immune system, leading to a reduction in the number and severity of PsA symptoms.
Effectiveness for psoriatic arthritis
Humira has shown to be effective in treating moderate to severe PsA in adults.
In clinical studies, researchers compared Humira with a placebo to see if Humira was better at reducing the severity of PsA.
In one study, 20% of people who took Humira had a 70% reduction in the severity and number of PsA symptoms after 12 weeks. This was compared with just 1% of people who took a placebo. After 24 weeks, 23% of people who took Humira had a 70% reduction in PsA symptoms compared with 1% of people who took a placebo.
Humira for uveitis
Humira is FDA-approved to treat certain types of uveitis in adults that aren’t caused by infection:
- intermediate uveitis: inflammation (swelling) of the middle part of the eye
- posterior uveitis: inflammation of the back of the eye
- panuveitis uveitis: inflammation of the entire eye
Uveitis refers to swelling of the middle layer of the eye, called the uvea. This causes symptoms such as pain, eye floaters, blurred vision, sensitivity to light, and redness in the eye. Left untreated, severe cases of uveitis can lead to vision loss.
Sometimes uveitis is the result of an infection. Other times, the condition is due to an autoimmune disease (a condition in which your immune system mistakenly attacks your body) such as rheumatoid arthritis or psoriasis.
Humira treats uveitis by blocking a protein called TNF. This decreases the activity of your immune system, leading to healing of damage to the eye and improved vision.
Effectiveness for uveitis
Humira has been proven effective for treating uveitis that isn’t due to an infection.
In clinical studies, researchers wanted to see how long Humira worked to treat uveitis in adults. The time at which uveitis symptoms worsened was called treatment failure. This is when uveitis lesions or symptoms get worse (either in number or severity) while a person is receiving treatment. People in the studies took either Humira or a placebo.
The researchers found that uveitis symptoms worsened in 39.1% to 54.5% of people who took Humira. This was compared with 55.0% to 78.5% of people who took a placebo.
In one study, uveitis symptoms worsened after 5.6 months in half of the people who received Humira. In comparison, uveitis symptoms worsened after 3 months in half of the people who received a placebo.
Study results in children
In a different study, researchers wanted to see how long it took for children between the ages of 2 and 17 years with uveitis to reach treatment failure. Children in the studies took either Humira with methotrexate or a placebo with methotrexate.
The researchers found that 26.7% of children who took Humira had treatment failure, compared with 60% of children who took a placebo. Less than half of the children who took Humira had reached treatment failure after 80 weeks. In comparison, it took half of the children who took a placebo 24.1 weeks to reach treatment failure.
Humira for ankylosing spondylitis (AS)
Humira is FDA-approved to treat ankylosing spondylitis that’s active in adults. “Active” means that you have symptoms. The goal of Humira is to reduce the signs and symptoms of AS.
AS is a form of arthritis that primarily affects your spine. It can also cause joint pain and stiffness in other parts of your body. AS results in severe inflammation (swelling) in your vertebrae, which are the small bones that form your spine. AS can lead to chronic (long-term) pain, disability, and spinal deformities in severe cases.
Sometimes AS can have periods of flare-ups. These are times when symptoms get worse. There can also be periods of remission. These are times when symptoms ease, become less frequent, or disappear.
It’s not entirely known what causes AS. It’s believed that incorrect immune system responses may play a role, meaning the immune system becomes active when it shouldn’t. Humira treats AS by blocking a protein called TNF. This decreases the activity of your immune system, leading to a reduction in AS symptoms and flare-ups.
Effectiveness for ankylosing spondylitis
Humira has been proven effective for treating AS in adults.
Clinical studies looked at whether Humira was better than a placebo at easing AS symptoms.
Researchers found that 58% of people who took Humira had a 20% improvement in AS symptoms at week 12. In comparison, 21% of people who took placebo had a 20% improvement in AS symptoms.
In addition, after 24 weeks, researchers found that 22% of people who took Humira had very low levels of disease activity. This was compared with just 6% of people who took a placebo. A low level of disease activity was defined as fewer than 20 points on a 100-point scale across four categories that measured symptom frequency and severity. Compared with people who took a placebo, people who took Humira reported that their back pain and inflammation eased much more in severity and frequency.
Humira for juvenile idiopathic arthritis (JIA)
Humira is FDA-approved to treat juvenile idiopathic arthritis that’s moderate to severe, polyarticular, and active in children ages 2 years and older. “Polyarticular” means that the condition affects more than one joint. And “active” refers to the fact that the child has symptoms.
JIA, which was previously known as juvenile rheumatoid arthritis, is the most common type of arthritis found in children. Children with JIA often have joint pain, swelling, and stiffness. Most cases are mild, but if left untreated, JIA will likely worsen and can lead to chronic (long-term) pain and joint damage.
It’s not known exactly what causes JIA, but it’s believed that it’s the result of an autoimmune disease. This is a type of disease in which your immune system attacks your body’s own cells by mistake. Humira treats JIA by blocking a protein called TNF. This decreases the activity of your immune system.
Effectiveness for juvenile idiopathic arthritis
Humira was studied in children ages 4 to 17 years with polyarticular JIA. All children in the study had previously tried other medications for JIA. In this clinical study, researchers wanted to see whether Humira was more effective at reducing the severity of JIA symptoms compared with a placebo.
At 48 weeks, they found that 43% children who took Humira had symptom flare-ups, compared with 71% of children who took a placebo. Also, 37% of children who took Humira and methotrexate had symptom flare-ups, compared with 65% of children who took a placebo and methotrexate.
Humira for other conditions
In addition to the uses listed above, Humira may be prescribed off-label for other conditions. Off-label drug use means using a drug for a purpose other than what it’s been approved for by the FDA. And you may wonder if Humira can be prescribed for certain other conditions. Below is information on other possible uses for Humira.
Humira for osteoarthritis (off-label use)
Humira isn’t approved by the FDA for treating osteoarthritis, a type of arthritis. But the drug may be prescribed off-label for this condition.
One
If you’d like to learn more about using Humira for osteoarthritis, talk with your doctor. They can recommend the best treatments for you.
Humira for sarcoidosis (off-label use)
Sarcoidosis is a condition in which inflamed (swollen) cells in your body build up in masses or nodules (abnormal growths) on various organs. It most commonly affects the lungs and lymph nodes.
Humira isn’t FDA-approved to treat sarcoidosis. But the drug may be prescribed off-label for this condition.
A systematic review looked at studies of TNF blocker drugs for treating different types of sarcoidosis. The review found that Humira may work to treat forms of sarcoidosis that affect the skin or eyes.
If you’re interested in learning more about taking Humira for sarcoidosis, talk with your doctor. They can recommend the best treatments for you.
Humira for IBS (not an appropriate use)
Irritable bowel syndrome (IBS) is a digestive disorder that can cause steady discomfort over the long term.
Humira isn’t FDA-approved to treat IBS.
If you’d like to learn about possible treatments for IBS, talk with your doctor.
Humira for lupus (may not be an appropriate use)
Lupus is a type of autoimmune disease (a condition in which your immune system attacks your body’s own cells by mistake).
It’s not known if Humira is effective for treating lupus. Some scientists believe that drugs called tumor necrosis factor (TNF) blockers may be useful in treating lupus. Humira is a type of TNF blocker.
On the other hand, Humira can cause an immune system reaction called lupus-like syndrome. (See the “Humira side effects” section above to learn more.) Although very rare, this was a reported side effect of the drug during clinical studies.
If you have questions about treatments for lupus, talk with your doctor.
Humira and children
The FDA has approved Humira to treat the following conditions in children:
- moderate to severe juvenile idiopathic arthritis in children ages 2 years and older
- moderate to severe Crohn’s disease in children ages 6 years and older
- moderate to severe ulcerative colitis in children ages 5 years and older
- certain types of uveitis in children ages 2 years and older
- moderate to severe hidradenitis suppurativa in children ages 12 years and older
For more information on the conditions Humira can be used to treat in children, refer to the individual sections above.
Depending on the condition it’s being used to treat, Humira may or may not be used with other drugs.
Humira is commonly used with a class of medications known as disease-modifying antirheumatic drugs (DMARDs).
Examples of DMARDs include:
For example, Humira is commonly used with methotrexate to treat rheumatoid arthritis (RA) and juvenile idiopathic arthritis (JIA).
If you have questions about taking Humira with other medications, talk with your doctor.
Humira comes in three forms: a prefilled pen, a prefilled syringe, and a vial of liquid solution. The drug is given as a subcutaneous injection. A healthcare professional can give you Humira injections. But you may be able to give yourself injections at home with the pen or syringe if your doctor approves. You’d need to be trained first.
Both the pens and syringes are prefilled with a single dose of Humira and come with needles. There are also special Humira “starter packs” available that contain several prefilled pens or syringes.
Humira also comes in a single-dose vial. But this form can only be used by healthcare professionals. You won’t be able to use vials yourself.
If you have questions about which form of Humira is right for you or how to give yourself injections, talk with your doctor.
Humira is given as a subcutaneous injection. You may be able to give yourself Humira injections at home. If so, your doctor or pharmacist will first show you how to use the medication. However, in some cases you may still need to visit your doctor’s office for the injections.
Humira injection sites
You can give yourself Humira injections in your belly or the front of your thigh. Be sure to inject the drug directly into your skin. Don’t try to inject Humira through your clothing.
And be sure to choose a different spot on your body each time you give yourself an injection. This new site should be at least 1 inch away from the last area you used. This helps prevent your skin from becoming too sensitive and reduces the risk of injection site reactions, like pain. Don’t inject Humira in a spot where your skin is red, bruised, tender, or hard.
For more information on how to properly give yourself a Humira injection, please visit the drug manufacturer’s website. It has detailed step-by-step video instructions.
When to take
Depending on the condition Humira is being used to treat, you’ll give yourself or receive an injection every 1 to 2 weeks. (For more about how often you’ll need injections, see the “Humira dosage” section above.)
To help make sure that you don’t miss a dose, try setting a reminder on your phone. You can also write your schedule in a calendar.
Other drugs are available that can treat your condition. Some may be a better fit for you than others. If you’re interested in finding an alternative to Humira, talk with your doctor. They can tell you about other medications that may work well for you.
Note: Some of the drugs listed below are used off-label to treat these specific conditions. Off-label drug use means using a drug for a purpose other than what it’s been approved for by the FDA.
Alternatives for plaque psoriasis
Examples of other drugs that may be used to treat plaque psoriasis include:
- topical (applied to the skin) treatments, such as:
- synthetic vitamin D
- topical corticosteroids
- topical retinoids
- certolizumab pegol (Cimzia)
- secukinumab (Cosentyx)
- etanercept (Enbrel)
- methotrexate (Trexall)
- apremilast (Otezla)
- infliximab (Remicade)
- ustekinumab (Stelara)
- ixekizumab (Taltz)
- guselkumab (Tremfya)
Alternatives for hidradenitis suppurativa (HS)
Examples of other drugs that may be used to treat hidradenitis suppurativa include:
- acetaminophen (Tylenol)
- corticosteroids, such as prednisone or prednisolone
- methotrexate (Trexall)
- nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen (Advil, Motrin)
- infliximab (Remicade)
- topical lidocaine
- topical retinoids
Alternatives for rheumatoid arthritis (RA)
Examples of other drugs that may be used to treat rheumatoid arthritis include:
- acetaminophen (Tylenol)
- certolizumab pegol (Cimzia)
- corticosteroids, such as prednisone or methylprednisolone
- etanercept (Enbrel)
- hydroxychloroquine (Plaquenil)
- leflunomide (Arava)
- methotrexate (Trexall)
- nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen (Advil, Motrin) or celecoxib (Celebrex)
- abatacept (Orencia)
- infliximab (Remicade)
- golimumab (Simponi)
- tofacitinib (Xeljanz)
Alternatives for psoriatic arthritis (PsA)
Examples of other drugs that may be used to treat psoriatic arthritis include:
- certolizumab pegol (Cimzia)
- secukinumab (Cosentyx)
- etanercept (Enbrel)
- leflunomide (Arava)
- nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen (Advil, Motrin) or celecoxib (Celebrex)
- abatacept (Orencia)
- apremilast (Otezla)
- infliximab (Remicade)
- golimumab (Simponi)
- ustekinumab (Stelara)
- ixekizumab (Taltz)
- tazarotene (Tazorac)
- tofacitinib (Xeljanz)
Alternatives for ulcerative colitis (UC)
Examples of other drugs that may be used to treat ulcerative colitis include:
- azathioprine (Imuran)
- balsalazide (Colazal)
- corticosteroids, such as budesonide, hydrocortisone, and prednisone
- vedolizumab (Entyvio)
- mesalamine (Asacol HD, Lialda)
- olsalazine (Dipentum)
- infliximab (Remicade)
- golimumab (Simponi)
- ustekinumab (Stelara)
- sulfasalazine (Azulfidine)
- tofacitinib (Xeljanz)
Alternatives for Crohn’s disease (CD)
Examples of other drugs that may be used to treat Crohn’s disease include:
- antibiotics, such as metronidazole or ciprofloxacin (Cipro)
- azathioprine (Imuran)
- balsalazide (Colazal)
- certolizumab pegol (Cimzia)
- corticosteroids, such as budesonide, hydrocortisone, and prednisone
- vedolizumab (Entyvio)
- mesalamine (Asacol HD, Lialda)
- methotrexate (Trexall)
- infliximab (Remicade)
- ustekinumab (Stelara)
Alternatives for uveitis
Examples of other drugs that may be used to treat uveitis include:
- azathioprine (Imuran)
- corticosteroids, such as prednisone
- cyclosporine (Gengraf, Sandimmune)
- methotrexate (Trexall)
- mycophenolate mofetil (CellCept)
- infliximab (Remicade)
- rituximab (Rituxan)
Alternatives for ankylosing spondylitis (AS)
Examples of other drugs that may be used to treat ankylosing spondylitis include:
- certolizumab pegol (Cimzia)
- corticosteroids, such as prednisone or prednisolone
- secukinumab (Cosentyx)
- etanercept (Enbrel)
- nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen (Advil, Motrin) or naproxen (Aleve)
- infliximab (Remicade)
- golimumab (Simponi)
- sulfasalazine (Azulfidine)
- ixekizumab (Taltz)
Alternatives for juvenile idiopathic arthritis (JIA)
Examples of other drugs that may be used to treat juvenile idiopathic arthritis include:
- tocilizumab (Actemra)
- etanercept (Enbrel)
- leflunomide (Arava)
- methotrexate (Trexall)
- nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen (Advil, Motrin) or naproxen (Aleve)
- abatacept (Orencia)
- rituximab (Rituxan)
- sulfasalazine (Azulfidine)
You may wonder how Humira compares with other medications that are prescribed for similar uses. Here we look at how Humira and Entyvio are alike and different.
Ingredients
Humira contains the active drug adalimumab. Entyvio contains the active drug vedolizumab. Both adalimumab and vedolizumab are types of medications called biologics. Biologics are medications made from living cells.
Both adalimumab and vedolizumab are kinds of medications known as monoclonal antibodies. These are a type of biologic medication made from immune system cells.
Humira belongs to a class of medications called tumor necrosis factor (TNF) blockers. Entyvio belongs to a class of medications called integrin receptor antagonists. A class of medications is a group of drugs that work in a similar way.
Uses
The Food and Drug Administration (FDA) has approved Humira to treat the following conditions:
- rheumatoid arthritis (RA) in adults
- juvenile idiopathic arthritis (JIA) in children ages 2 years and older
- psoriatic arthritis (PsA) in adults
- ankylosing spondylitis (AS) in adults
- Crohn’s disease (CD) in adults as well as children 6 years and older
- ulcerative colitis (UC) in adults as well as children 5 years and older
- plaque psoriasis in adults
- hidradenitis suppurativa (HS) in adults as well as children ages 12 years and older
- certain types of uveitis in adults as well as children ages 2 years and older
For details on how Humira is used to treat these conditions, see the sections “Humira for plaque psoriasis” and “Other uses for Humira” above.
Entyvio is approved by the FDA to treat moderate to severe UC or CD, in adults.
So both Humira and Entyvio are FDA-approved to treat moderate to severe UC in adults and moderate to severe CD in adults. Humira can also be used to treat UC and CD in certain children.
Drug forms and administration
Humira comes in three forms: a single-dose pen, a single-dose prefilled syringe, and a single-dose vial of liquid solution.
Humira is given as a subcutaneous injection. A healthcare professional can give you Humira injections. But you may be able to give yourself injections at home with the pen or syringe if your doctor approves. You’d need to be trained first. The vial form of Humira can be used only by healthcare professional. You won’t be able to use vials yourself.
Entyvio comes as a powder in a single-dose vial. A healthcare professional will add a liquid to it to make a solution. The drug is given as an IV infusion. Entyvio must be given by a healthcare professional.
Side effects and risks
Although Humira and Entyvio contain different active drugs, these medications can cause very similar side effects, but some different ones as well. Below are examples of these side effects.
Mild side effects
These lists contain up to 10 of the most common mild side effects that can occur with Humira, with Entyvio, or with both Humira and Entyvio (when taken individually).
- Can occur with Humira:
- injection site reactions (itching, pain, or swelling near the injection spot)
- Can occur with Entyvio:
- Can occur with both Humira and Entyvio:
- upper respiratory infections, such as the common cold
- rash
Serious side effects
These lists contain examples of serious side effects that can occur with Humira, with Entyvio, or with both drugs (when taken individually).
- Can occur with Humira:
- lupus-like syndrome (an immune system reaction)
- nerve disorders or demyelinating diseases, such as seizures or multiple sclerosis (MS)
- blood disorders, such as anemia (low level of red blood cells)
- Can occur with Entyvio:
- infusion-related reactions, such as nausea or fever
- a rare, aggressive viral disease known as progressive multifocal leukoencephalopathy
- Can occur with both Humira and Entyvio:
- liver damage
- risk of serious infections,* such as tuberculosis (TB) or pneumonia
* Humira has
Effectiveness
The only conditions both Humira and Entyvio are used to treat are ulcerative colitis (UC) and Crohn’s disease (CD) in adults. Humira can also be used to treat UC in children ages 5 years and older and CD in children ages 6 years and older.
Effectiveness in treating ulcerative colitis
The use of Humira and Entyvio in treating moderate to severe UC in adults has been directly compared in a clinical study. Researchers randomly assigned 769 adults to receive either Humira or Entyvio over 52 weeks, and then analyzed the results. The goal was to see which drug was better at causing clinical remission of UC. This is a reduction in inflammation (swelling), which leads to fewer and less severe symptoms.
After 52 weeks, 31.3% of people who received Entyvio were in clinical remission compared to 22.5% of people who received Humira. However, 21.8% of people who received Humira were able to achieve remission without using corticosteroids, compared with 12.6% of people who received Entyvio.
To find out how effective these drugs are in children, see the prescribing information for Humira and Entyvio.
Effectiveness in treating Crohn’s disease
A different analysis of clinical studies looked at people with moderate to severe Crohn’s disease. Researchers found that Humira was more likely to cause remission than a placebo. Entyvio, on the other hand, wasn’t more likely to cause remission than a placebo. The researchers also determined that Humira was more effective at keeping the disease in remission.
Costs
Humira and Entyvio are both brand-name drugs. The Food and Drug Administration (FDA) has approved seven biosimilar versions of Humira: Hyrimoz, Hadlima, Amjevita, Cyltezo, Abrilada, Hulio, and Yusimry. However, these biosimilars are not yet available for use.
No biosimilars of Entyvio are currently available, but they are in development.
A biosimilar is a medication that’s similar to a brand-name drug. A generic medication, on the other hand, is an exact copy of a brand-name drug. Biosimilars are based on biologic medications, which are created from parts of living organisms. Generics are based on regular drugs made from chemicals. Biosimilars and generics also tend to cost less than brand-name medications.
Humira typically costs more than Entyvio over the course of 1 year. The actual price you’ll pay for either drug depends on your insurance plan and your location.
You may wonder how Humira compares with other medications that are prescribed for similar uses. Here we look at how Humira and Cosentyx are alike and different.
Ingredients
Humira contains the active drug adalimumab. Cosentyx contains the active drug secukinumab. Both adalimumab and secukinumab are types of medications called biologics. Biologics are medications made from living cells.
Both adalimumab and secukinumab are kinds of medications known as monoclonal antibodies. These are a type of biologic medication made from immune system cells.
Humira belongs to a class of medications called tumor necrosis factor (TNF) blockers. Cosentyx belongs to a class of medications called interleukin-17A (IL-17A) blockers. A class of medications is a group of drugs that work in a similar way.
Uses
The Food and Drug Administration (FDA) has approved Humira to treat the following conditions:
- rheumatoid arthritis (RA) in adults
- juvenile idiopathic arthritis (JIA) in children ages 2 years and older
- psoriatic arthritis (PsA) in adults
- ankylosing spondylitis (AS) in adults
- Crohn’s disease (CD) in adults as well as children 6 years and older
- ulcerative colitis (UC) in adults as well as children 5 years and older
- plaque psoriasis in adults
- hidradenitis suppurativa (HS) in adults as well as children ages 12 years and older
- certain types of uveitis in adults as well as children ages 2 years and older
For details on how Humira is used to treat these conditions, see the sections “Humira for plaque psoriasis” and “Other uses for Humira” above.
Cosentyx is FDA-approved to treat:
- Plaque psoriasis that’s moderate to severe in adults and children 6 years and older. To use Cosentyx, you must be eligible for phototherapy or systemic therapy. Phototherapy uses light to treat plaque psoriasis. And systemic therapy refers to medications that work through your entire body to help stop plaques (patches on your skin) from forming.
- Psoriatic arthritis in adults and children 2 years and older. The psoriatic arthritis must be active. “Active” means that you currently have symptoms.
- Ankylosing spondylitis in adults. The ankylosing spondylitis must be active.
- Non-radiographic axial spondyloarthritis in adults. The non-radiographic axial spondyloarthritis must be active.
- Arthritis related to enthesitis in adults and children 4 years and older. The enthesis is the area where ligaments and tendons attach to bones. Arthritis related to enthesitis must be active.
So, both Humira and Cosentyx are approved to treat plaque psoriasis in adults and certain children, psoriatic arthritis in adults and certain children, and ankylosing spondylitis in adults.
Drug forms and administration
Humira comes in three different forms: a single-dose pen, a single-dose prefilled syringe, and a single-dose vial of liquid solution.
Cosentyx also comes in three different forms: a single-use Sensoready pen, a single-use prefilled syringe, and a single-use vial of powder. A healthcare professional will add a liquid to it to make a solution.
Both Humira and Cosentyx are given by subcutaneous injection. A healthcare professional can give you the injections. But you may be able to give yourself injections at home with the pen or syringe if your doctor approves. You’d need to be trained first. The vial forms of the drugs can be used only by healthcare professionals. You won’t be able to use vials yourself.
Side effects and risks
Humira and Cosentyx contain different but similar active drugs. Therefore, these medications can cause very similar side effects, but some different ones as well. Below are examples of these side effects.
Mild side effects
These lists contain up to 10 of the most common mild side effects that can occur with Humira, with Cosentyx, or with both Humira and Cosentyx (when taken individually).
- Can occur with Humira:
- rash
- injection site reactions (itching, pain, or swelling near the injection spot)
- Can occur with Cosentyx:
- Can occur with both Humira and Cosentyx:
- headache
- upper respiratory infections, such as the common cold
Serious side effects
These lists contain examples of serious side effects that can occur with Humira, with Cosentyx, or with both drugs (when taken individually).
- Can occur with Humira:
- lupus-like syndrome (an immune system reaction)
- nerve disorders or demyelinating diseases, such as seizures or multiple sclerosis (MS)
- blood disorders, such as anemia (low level of red blood cells)
- risk of cancer,* such as lymphoma (cancer of the white blood cells)
- liver damage
- Can occur with Cosentyx:
- new or worsening inflammatory bowel disease
- Can occur with both Humira and Cosentyx:
- risk of serious infections,* such as tuberculosis (TB) and pneumonia
* Humira has
Effectiveness
Humira and Cosentyx have different FDA-approved uses, but they’re both used to treat plaque psoriasis, psoriatic arthritis, and ankylosing spondylitis.
Studies of Humira and Cosentyx were compared in a large review of studies. The researchers found that Cosentyx was more effective than Humira at treating plaque psoriasis, psoriatic arthritis, and ankylosing spondylitis.
A recent
Another study is currently comparing Humira and Cosentyx for ankylosing spondylitis treatment in adults. Results from this study will be made available soon.
Costs
Humira and Cosentyx are both brand-name drugs. The Food and Drug Administration (FDA) has approved seven biosimilar versions of Humira: Hyrimoz, Hadlima, Amjevita, Cyltezo, Abrilada, Hulio, and Yusimry. However, these biosimilars are not yet available for use.
Cosentyx doesn’t come in biosimilar form.
A biosimilar is a medication that’s similar to a brand-name drug. A generic medication, on the other hand, is an exact copy of a brand-name drug. Biosimilars are based on biologic medications, which are created from parts of living organisms. Generics are based on regular drugs made from chemicals. Biosimilars and generics also tend to cost less than brand-name medications.
Humira and Cosentyx costs will vary depending on the form and dose your doctor prescribes. The actual price also depends on your insurance plan, your location, and the pharmacy you use.
You may wonder how Humira compares with Remicade. These drugs are used to treat many of the same conditions. Check out this article to learn how these drugs compare. Also, be sure to talk with your doctor about which drug is right for you.
There are no known interactions between alcohol and Humira at this time. According to the drug’s manufacturer, there aren’t any risks linked to drinking alcohol during treatment with Humira. But, there aren’t any studies of how alcohol use might impact Humira treatment.
However, drinking alcohol can affect some of the conditions Humira is used to treat. For example, alcohol may worsen symptoms in some people with ulcerative colitis (UC) or Crohn’s disease (CD). Alcohol may also affect people with plaque psoriasis by increasing inflammation (swelling), which can cause plaques (patches on skin) to form.
If you have questions about drinking alcohol while using Humira, talk with your doctor.
Humira can interact with several other medications.
Different interactions can cause different effects. For instance, some interactions can interfere with how well a drug works. Other interactions can increase the number of side effects or make them more severe.
Humira and other medications
Below is a list of medications that can interact with Humira. This list doesn’t contain all drugs that may interact with Humira.
Before taking Humira, talk with your doctor and pharmacist. Tell them about all prescription, over-the-counter, and other drugs you take. Also tell them about any vitamins, herbs, and supplements you use. Sharing this information can help you avoid potential interactions.
If you have questions about drug interactions that may affect you, ask your doctor or pharmacist.
Humira and drugs that weaken your immune system
Taking Humira with certain medications that weaken your immune system can increase your risk for serious infections.
Humira has a
Disease-modifying antirheumatic drugs
In people with rheumatoid arthritis (RA), certain medications called disease-modifying antirheumatic drugs (DMARDs) are often used to slow down the progression of RA and relieve symptoms. Some DMARDs are biologic drugs, like Humira. Biologics are medications made from living cells. These biologic DMARDs, when used together with Humira, can weaken your immune system. This makes you more likely to develop serious infections.
Using biologic DMARDs, such as abatacept (Orencia) and anakinra (Kineret), isn’t recommended when you’re taking Humira.
However, it’s fine to take Humira with a DMARD called methotrexate (Trexall), even if you have RA. According to Humira’s manufacturer, these two drugs can be safely used together.
Tumor necrosis factor blockers
Humira also shouldn’t be used with drugs called tumor necrosis factor (TNF) blockers for the same reason. TNF blockers can weaken your immune system. Because Humira is a TNF blocker, taking it with other TNF blockers can put you at risk for serious infections.
Examples of TNF blockers include:
If you’re taking a drug that can weaken your immune system, or if you’re not sure, talk with your doctor before starting Humira treatment. They can advise you on the right medications to use.
Humira and certain drugs with a narrow safe range
Humira can affect the way your body digests certain medications. For most drugs, this effect isn’t strong enough that you would notice it in any way.
However, some medications have a narrow safe range. This means that even small changes in the amount of drug in your body can cause the drug to not work or cause you to have side effects. Using Humira with these medications may result in you having harmful side effects from either drug. (For side effects of Humira, see the “Humira side effects” section above.)
Examples of medications with a narrow safe range include:
- warfarin (Jantoven)
- cyclosporine (Gengraf, Sandimmune)
- Theophylline (Theo-24)
Before starting Humira treatment, be sure that your doctor and pharmacist are aware of all the medications you’re taking. They’ll work with you to make sure Humira is safe for you to use with your medications.
Humira and vaccines
While taking Humira, you shouldn’t get any live vaccines. Live vaccines contain a weakened version of the virus or bacterium they’re meant to treat. If you have a healthy immune system, live vaccines usually don’t cause infections. But if your immune system is weak due to taking Humira, getting a live vaccine may cause an infection.*
Examples of live vaccines include:
- measles, mumps, rubella (MMR)
- nasal spray form of the flu vaccine (FluMist)
- oral form of the typhoid vaccine (Vivotif)
- chickenpox (Varivax)
- shingles vaccine (Shingrix)
Before you start taking Humira, ask your doctor if your vaccines are up to date. They may recommend that you get certain vaccines before your Humira treatment begins.
* Humira has
Humira and herbs and supplements
There aren’t any herbs or supplements that have been specifically reported to interact with Humira. However, you should still check with your doctor or pharmacist before using any of these products while taking Humira.
Humira and foods
There aren’t any foods that have been specifically reported to interact with Humira. If you have any questions about eating certain foods with Humira, talk with your doctor.
Here are answers to some frequently asked questions about Humira.
Can I stop using Humira and then restart it later?
Yes. As long as you follow your doctor’s instructions, you can stop taking Humira and then restart your treatment later on. But the drug may not work as well the second time.
This is because it’s possible for your body to form antibodies against Humira. (Antibodies are immune system proteins that attack substances, such as Humira, and can keep them from working well.) So when you stop and then restart your treatment, the antibodies can make the drug less effective.
However, one study looked at people with rheumatoid arthritis (RA) who stopped taking Humira, and then started their treatment again. These people had a flare-up (worsening of symptoms) after they ended their treatment. But taking Humira the second time was effective at easing the RA flare-up within 9 months in 100% of the people. Note that this was a small study, and further research is still needed.
It’s important that you don’t stop your Humira treatment without first checking with your doctor. They can help answer any questions you have.
Is Humira ever given as an infusion?
No, Humira is given as a subcutaneous injection (an injection under your skin). An IV infusion is when a drug is injected directly into a vein over a period of time. Infusions must be given by healthcare professionals.
With Humira, a healthcare professional can give you the injections. But you may be able to give yourself injections at home with the pen or syringe forms if your doctor approves. You’d need to be trained first. This way, you wouldn’t have to go to your doctor’s office to receive your doses.
If you have any questions about how to take Humira, talk with your doctor.
Will I have withdrawal symptoms if I stop using Humira?
Humira itself isn’t likely to cause withdrawal symptoms. (Withdrawal refers to symptoms that can occur if you stop taking a drug that your body is dependent on.) However, if you stop using Humira, there’s a strong possibility that symptoms of your condition will return or worsen.
For example, in clinical studies, 22% of people with hidradenitis suppurativa (HS) who stopped taking Humira had a flare-up of their HS symptoms after they ended their treatment.
If you have questions about ending your use of Humira, talk with your doctor. You shouldn’t stop taking the drug on your own.
Is it safe to have surgery while I’m taking Humira?
There’s not a lot of research on whether it’s safe to have surgery while taking Humira.
One potential side effect of Humira is an increased risk of serious infections,* such as tuberculosis (TB). This is because Humira can weaken your immune system. Having surgery also places you at a higher risk for infection. So there’s concern that having surgery while taking Humira can raise your risk further.
In a
If you plan on having surgery while taking Humira, talk with your doctor. They’ll help determine the right treatment plan for you.
* Humira has boxed warnings for risk of serious infections. A boxed warning is the most serious warning from the Food and Drug Administration (FDA). For more information, see “FDA warnings” at the beginning of this article.
What blood tests will I need before or during Humira treatment?
You’ll need several tests before starting Humira treatment and while you take the drug, including:
- Tuberculosis (TB). Before you start taking Humira, your doctor will check you for TB. While you take the medication, they’ll monitor you for any symptoms of infection. This is because Humira can weaken your immune system and make you more likely to develop an infection.*
- Hepatitis B. Your doctor will also want to see if you have the hepatitis B virus (HBV). Humira can cause HBV to become reactivated in people who previously had active HBV. (“Active” means that you have symptoms.) Your doctor may keep testing you for HBV while you take Humira and for several months after you stop your treatment.
- Liver function. While you take Humira, your doctor will likely order blood tests to make sure your liver is working properly. Humira can cause liver damage and sudden liver failure in rare cases.
- Humira level. During your treatment with Humira, your doctor may also order lab tests to check the level of Humira in your blood. Although more research is needed, keeping Humira levels in the blood within a specific range may be ideal for treatment.
- Humira antibodies. In addition, your doctor may order a blood test that checks for antibodies to Humira. (For more about antibodies, see “Can I stop using Humira and then restart it later?” above.) People who form antibodies against Humira are less likely to respond well to the drug.
If you have questions about what tests you’ll need before and during your Humira treatment, check with your doctor.
* Humira has
How will I know if Humira isn’t working for my condition?
You’ll know Humira isn’t working for your condition if your symptoms don’t ease or ease so little that your daily life is still affected by your condition. If Humira is working, you should notice symptom relief within the first few months of taking the drug.
If you have questions about how well Humira is working for you, talk with your doctor.
The
In one study, Humira was used to treat rheumatoid arthritis (RA) or Crohn’s disease (CD) during pregnancy. Major congenital anomalies (commonly known as birth defects) occurred in 10% of pregnant females* who took Humira, compared with 7.5% of females who didn’t take the drug.
Due to the small number of people in the study, these results don’t provide enough information to state for certain that Humira causes major congenital anomalies. This is because factors other than Humira between the two groups could lead to the differences in the rate of congenital anomalies.
If you’re pregnant or plan on becoming pregnant, talk with your doctor before taking Humira. They’ll review the risks and benefits of the drug with you.
* Sex and gender exist on spectrums. Use of the term “female” in this article refers to sex assigned at birth.
It’s not known if Humira is safe to take during pregnancy. If you’re sexually active and you or your partner can become pregnant, talk with your doctor about your birth control needs while you’re using Humira.
You should not breastfeed while taking Humira, according to the drug’s manufacturer.
Humira passes into human breast milk and can be transferred to a breastfed child. However, there isn’t enough data to know whether the amount is safe or if it could potentially harm the child. More research is still needed.
If you’re breastfeeding or are planning to, talk with your doctor. They can explain the best ways to feed your child and what treatment options are available to you.
When you get Humira from the pharmacy, the pharmacist will add an expiration date to the label on the carton. This date is typically 1 year from the date they dispensed the medication.
The expiration date helps guarantee that the medication is effective during this time. The
Storage
How long a medication remains good can depend on many factors, including how and where you store the medication.
You should keep Humira in the original carton in a refrigerator at 36°F to 46°F (2°C to 8°C). This helps protect the drug from light. Never freeze Humira.
If necessary (such as when traveling), you can keep Humira at room temperature to a maximum of 77°F (25°C) for up to 14 days. Be sure to protect the drug from light at all times until you give yourself your dose.
Disposal
If you no longer need to take Humira and have leftover medication, it’s important to dispose of it safely. This helps prevent others, including children and pets, from taking the drug by accident. It also helps keep the drug from harming the environment.
This article provides several useful tips on medication disposal. You can also ask your pharmacist for information on how to dispose of your medication.
When you have an autoimmune disease, your immune system mistakenly sees something in your body as a threat and attacks it. This leads to the problems and symptoms seen in conditions such as plaque psoriasis, rheumatoid arthritis (RA), Crohn’s disease (CD), and psoriatic arthritis (PsA).
It’s not exactly known what causes autoimmune diseases. Experts think that a protein called tumor necrosis factor (TNF) is a major contributor to inflammation (swelling and damage) in these conditions. When the body sees a threat, TNF triggers inflammation to help fight the threat. But when TNF incorrectly sees the body as a threat, TNF’s response can lead to healthy tissue being damaged. Inflammation can also lead to pain.
Humira works by binding to TNF and blocking it from causing an inflammatory reaction. This reduces inflammation in people with autoimmune disease.
How long does it take to work?
According to the manufacturer of Humira, some people see benefits as soon as 2 weeks after starting treatment. But the manufacturer also notes that some people may need to take Humira for 3 months or longer before they notice their symptoms ease.
This drug comes with several precautions.
FDA warnings
This drug has
Risk of serious infections
Taking Humira can increase your risk for serious infections that may lead to a hospital stay or death. These infections include sepsis (a severe response to an infection, which may lead to severe problems, including death), a kind of lung infection called tuberculosis (TB), and histoplasmosis (a fungal infection similar to pneumonia). They also include infections that usually affect people with weakened immune systems.
During your treatment, your doctor will monitor you closely for signs of an infection. If you do develop a serious infection, they’ll likely have you stop taking Humira. Before you start using Humira, your doctor will test you for TB. If you have TB, it’ll need to be treated before you begin Humira treatment.
Risk of cancer
Lymphomas (cancers of the white blood cells) and other cancers have occurred in people who took Humira, including children and teens. Some of these cancers were fatal.
A rare type of lymphoma called hepatosplenic T-cell lymphoma occurred mostly in young adult and teen males* who had ulcerative colitis (UC) or Crohn’s disease (CD). These two conditions affect the digestive system. Hepatosplenic T-cell lymphoma is a rare form of cancer that affects cells in your liver and spleen.
* Sex and gender exist on spectrums. Use of the term “male” in this article refers to sex assigned at birth.
Other precautions
Before taking Humira, talk with your doctor about your health history. Humira may not be right for you if you have certain medical conditions or other factors affecting your health. These include:
- Heart failure. Humira is a type of drug called a tumor necrosis factor (TNF) blocker. TNF blockers such as Humira have been reported to cause heart failure to worsen. So if you have heart failure, your doctor should monitor you closely during your Humira treatment.
- Nervous system reactions. TNF blockers such as Humira can worsen symptoms of nerve disorders and demyelinating diseases. Examples of these kind of diseases are seizures and multiple sclerosis (MS). If you have a nerve disorder or demyelinating disease, ask your doctor if Humira is the best choice for you.
- Hepatitis B. Humira may cause the hepatitis B virus to reactivate. This means that if you had a hepatitis B infection in the past but no longer have symptoms, taking Humira may cause you to have symptoms again. If you’ve ever had hepatitis B, your doctor should closely monitor you during your Humira treatment.
- Cancer. TNF blockers such as Humira can worsen tumors (masses of cancerous tissue) or cancers. So if you have a tumor or cancer, your doctor may recommend a medication other than Humira.
- Liver damage. Severe liver damage has been reported in people who use TNF blockers, including Humira. Talk with your doctor about any history of liver damage or liver disease you have. They may monitor you for signs and symptoms of worsening liver function during your Humira treatment.
- Active infection. You shouldn’t take Humira if you have an active infection. (“Active” means that you currently have symptoms.) Humira can weaken your immune system, which can worsen infections. Your doctor will likely treat the infection before you can start taking Humira.
- Allergy to latex or rubber. Some Humira products use a needle cover that may contain natural rubber latex. So if you have a latex allergy or a rubber allergy, tell your doctor before using Humira. They’ll likely recommend a different form of the drug that doesn’t contain latex or rubber.
- Allergy to Humira. If you’re allergic to Humira or any of its ingredients, you shouldn’t take the drug. Ask your doctor what other treatments are better choices for you.
- Pregnancy. It isn’t known whether Humira is safe for use during pregnancy. For more information, please see the “Humira and pregnancy” section above.
- Breastfeeding. You should not breastfeed while taking Humira. For more information, please see the “Humira and breastfeeding” section above.
Note: For more information about the potential negative effects of Humira, see the “Humira side effects” section above.
Using more than the recommended dosage of Humira can lead to serious side effects.
What to do in case of overdose
If you think you’ve taken or received too much of this drug, call your doctor. You can also call the American Association of Poison Control Centers at 800-222-1222 or use their online tool. But if your symptoms are severe, call 911 or go to the nearest emergency room right away.
The following information is provided for clinicians and other healthcare professionals.
Indications
Humira is approved by the Food and Drug Administration (FDA) to treat:
- moderate to severe rheumatoid arthritis (RA) in adults
- moderate to severe polyarticular juvenile idiopathic arthritis (JIA) in children ages 2 years and older
- psoriatic arthritis (PsA) in adults
- ankylosing spondylitis (AS) in adults
- moderate to severe Crohn’s disease (CD) in adults and in children ages 6 years and older
- moderate to severe ulcerative colitis (UC) in adults and in children ages 5 years and older
- moderate to severe chronic plaque psoriasis in adults who could use either systemic therapy or phototherapy, when other systemic therapies are less appropriate
- moderate to severe hidradenitis suppurativa (HS) in adults as well as children ages 12 years and older
- intermediate uveitis, posterior uveitis, and panuveitis that’s not caused by infection in adults as well as children ages 2 years and older
Mechanism of action
Humira directly binds to tumor necrosis factor (TNF)-alpha, blocking it from interacting with p55 and p75 TNF receptors on the cell surface. Biological responses induced or regulated by TNF are modulated by Humira, including reductions in the number of TNF molecules that cause leukocyte migration.
TNF is involved with immune-mediated and inflammation responses. Higher levels of TNF are also found in people with autoimmune diseases such as RA, AS, and PsA. It is unknown exactly how Humira treats these conditions, but it is believed to be related to its binding to TNF.
Pharmacokinetics and metabolism
Following subcutaneous administration, Humira takes approximately 131 hours to reach maximal concentration. The average terminal half-life is about 2 weeks (range: 10 to 20 days).
Contraindications
There are no absolute contraindications to Humira. Do not use Humira in patients allergic to adalimumab or to any ingredients of the drug product.
Storage
Humira should be refrigerated at 36°F to 46°F (2°C to 8°C) in the original carton. Humira should be kept in the original carton until administration in order to protect from light. Under no circumstances should Humira be frozen.
If necessary (such as when traveling), Humira may be kept at room temperature, to a maximum of 77°F (25°C), for up to 14 days.
Disclaimer: Medical News Today has made every effort to make certain that all information is factually correct, comprehensive, and up to date. However, this article should not be used as a substitute for the knowledge and expertise of a licensed healthcare professional. You should always consult your doctor or another healthcare professional before taking any medication. The drug information contained herein is subject to change and is not intended to cover all possible uses, directions, precautions, warnings, drug interactions, allergic reactions, or adverse effects. The absence of warnings or other information for a given drug does not indicate that the drug or drug combination is safe, effective, or appropriate for all patients or all specific uses.